Entrepreneur
Can a digital coach truly support menopause in the workplace?

By Gail MacLeitch, psychotherapist and VP of AI communications provider QuickBlox, and Leslie Taylor, co-founder of Half the Sky, the team behind BestYet—a digital platform which helps women take control of their menopausal journey.
Despite spending more than a third of their lives in menopause, most women face this transition with little support, personally or professionally. Instead, women tend to navigate menopause alone, often unclear on the full extent of what to expect, or when to expect it, until it arrives.
In 2024, nearly one-third of women said menopause impacted their job performance. Women reported reducing their work hours, turning down promotions, and quitting as outcomes of their symptoms. These numbers represent daily struggles playing out in the workplace.
Companies in 18 U.S. states, including Washington, California, Colorado, and New York, already offer their employees mandatory paid sick leave, but women have reported being penalized for using this time to cope with the side effects of menopause. Even in states with progressive leave policies, cultural stigma and managerial bias undermine their intended protection.
A UK study found that only a quarter of women taking sick leave felt able to tell their managers the real reason for their absence. While many wanted to protect their privacy, 34% said they were embarrassed, and another 32% said an unsupportive manager prevented them from speaking up.
Many of the symptoms women struggle with, such as self-doubt, low patience, heightened stress, and difficulty sleeping, can be helped with the right support.
Understanding Personal Symptoms
According to a survey of over 2,000 menopausal women in the UK, 84% experienced trouble sleeping, 73% experienced ‘brain fog’, and 69% experienced difficulties with anxiety or depression during menopause. However, the scale at which symptoms impact women varies and evolves over time.
What symptoms are your employees experiencing? How are these side effects influenced by controllable aspects of their lives, such as diet, exercise, and medication?
Companies can empower women with the tools to easily track and monitor their menopausal symptoms with secure and private daily logs. These tools help surface patterns in fatigue, mood, and concentration, correlated with sleep, food, and stress. Apps such as Health and Her and MENO already demonstrate similar solutions, providing individualized recommendations, such as meal guidance, mindfulness, and wellness nudges, based on symptom tracking and progress.
Gaining Confidential Guidance
Many women-centric midlife and menopause platforms are becoming available. Women who have used these report feeling more knowledgeable, confident, and in control of their menopause journey. The apps encourage self-reflection and build confidence around menopause discussions.
However, while menopause-at-work policies are becoming more common, conversations about accommodations remain deeply personal. Employers and managers should create safe pathways for private check-ins. Digital tools can help prep both parties with the right frameworks.
For example, a well-designed menopause support app might include pre-meeting reminders to ensure respectful, informed dialogue. Building guidance on how to handle disclosures and requests into the manager’s app user journey helps guarantee they see the right information when they need it. As menopause policies increasingly become mandatory, automated summaries that record what was discussed, what was agreed, and how confidentiality will be upheld provide a transparent data trail to protect both parties.
Companies can go a step further by using secure, no-code chatbot builders to provide 24/7 support. These tools, trained on medical research, HR policy, and expert guidance, can offer calm, evidence-based responses. When used responsibly, AI-enhanced chat tools become a supplemental layer of support and enable users to navigate to other pertinent workplace benefits and providers.
Building a Community
A problem shared is a problem halved, and this is especially true for menopause.
Women benefit from sharing what works and what doesn’t, from diet to hormone replacement therapy (HRT) to mindset. But often, just feeling understood can be transformative, especially in an isolating workplace context.
Features companies must consider for their female employees include:
- Community support: A private, judgment-free space to connect with other women navigating menopause, share stories, and exchange tips that actually work. Women can gain emotional and practical support by engaging in forum discussions.
- Daily prompts: Affirmations, meal suggestions, lifestyle tips, and wellness nudges that spark engagement.
- HR integration: Integrated menopause apps can give HR anonymized data on employee well-being and resource use. No or low-code tools can be embedded directly with no technical expertise, meaning HR teams can launch a wellness resource tailored to their workforce with minimal effort.
This support should extend across the organization. Menopause literacy for male employees, team leaders, and executives reduces stigma. Including resources and workshops directed at the company as a whole will help lead to a more empathetic workforce.
So, can a digital coach truly support menopause in the workplace? Yes, when it’s designed with empathy, grounded in science, and built to empower. The right digital tools don’t just track symptoms, they help women advocate for themselves, stay engaged, and thrive in their roles. The companies that design for midlife now won’t just support women. They’ll set the standard for what inclusive, intelligent, and future-ready health tech in the workplace can be.
Entrepreneur
Sun Pharma to acquire Organon in US$11bn deal
News
Women’s digital health market set to reach US$5.28 billion in 2026 – report
Entrepreneur
Future Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

Future Fertility Inc. has announced the closing of a US$4.1 million Series A financing round.
The round was led by M Ventures (the corporate venture capital arm of Merck KGaA, Darmstadt, Germany) and Whitecap Venture Partners, with participation from new investors Sandpiper Ventures, Gaingels, and Jolt VC.
The financing will accelerate Future Fertility’s commercial expansion into Asia-Pacific and support its entry into the United States, including planned FDA 510(k) clearance for additional products as part of a broader U.S. market entry strategy.
Proceeds will also advance the development of a broader AI platform, from egg assessment through to embryo transfer, designed to support clinicians, embryologists, and patients across the full IVF journey.
M Ventures and Whitecap have supported Future Fertility’s mission to translate AI innovation into meaningful clinical outcomes since the company’s earliest stages.
Oliver Hardick, investment director, M Ventures, said: “Future Fertility is addressing a critical unmet need in reproductive medicine with a differentiated AI platform grounded in clinical data and real-world workflow integration.
“We are excited to continue supporting the company and team because we believe its technology has the potential to improve decision-making for clinicians, bring greater clarity to patients, and help advance a more personalised standard of care in fertility treatment.”
Future Fertility’s AI platform addresses a long-standing gap in fertility care: historically, there has been no objective, clinically validated method for assessing egg quality (Gardner et al., 2025), despite it being one of the most important drivers of reproductive success.
The company’s suite of deep learning tools includes VIOLET™, MAGENTA™, and ROSE™, purpose-built for egg freezing, IVF, and egg donation respectively.
The tools are based on AI models trained and validated on more than 650,000 oocyte images and are deployed in over 300 clinics across 35 countries.
Rhiannon Davies, founding and managing partner, Sandpiper Ventures, said: “The best outcomes in fertility care globally come from better data and smarter tools. Future Fertility understands that, and they’ve built a platform that delivers on it.
“Sandpiper is proud to back a team turning rigorous science into real results for patients and clinicians alike.”
Partnerships with the world’s leading fertility networks – including IVI RMA and Eugin Group across Latin America and Europe, FertGroup Medicina Reproductiva in Brazil, and most recently announced Kato Ladies Clinic in Japan – reflect growing demand for objective, AI-powered oocyte assessment in fertility care. In the United States, ROSE™ is newly available under an FDA 513(g) determination.
Research shows that approximately 50 per cent of IVF patients do not understand their likelihood of success, and many discontinue treatment prematurely, even though cumulative success rates improve significantly with multiple cycles (McMahon et al., 2024).
By delivering earlier clarity on egg quality, Future Fertility’s tools support more informed conversations between clinicians and patients, helping set realistic expectations and guide decisions about next steps.
Future Fertility’s growing evidence base spans seven peer-reviewed publications in Human Reproduction, Reproductive BioMedicine Online, Fertility & Sterility, and Nature’s Scientific Reports, and more than 70 scientific abstracts accepted and presented with partner clinics at conferences worldwide.
Christine Prada, CEO, Future Fertility, said: “Fertility treatment is one of the most emotionally and physically demanding experiences a person can go through.
“Every patient deserves objective data, not just a best guess, to support better decisions at critical moments in their care.
“This funding means we can bring that clarity to more patients, in more countries, at a moment when it matters most.”
Find out more about Future Fertility at futurefertility.com

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
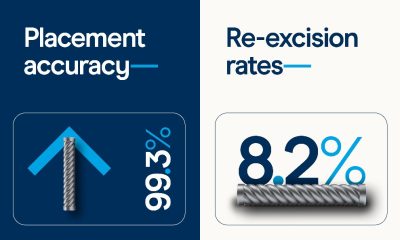
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy4 weeks ago
Pregnancy4 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Mental health4 weeks ago
Mental health4 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News4 weeks ago
News4 weeks agoResistance training has preventative effects in menopause, study finds

 Events2 weeks ago
Events2 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now