Hormonal health
Understanding the contraception crisis
How the one-size-fits-all approach fails women and why it has to change
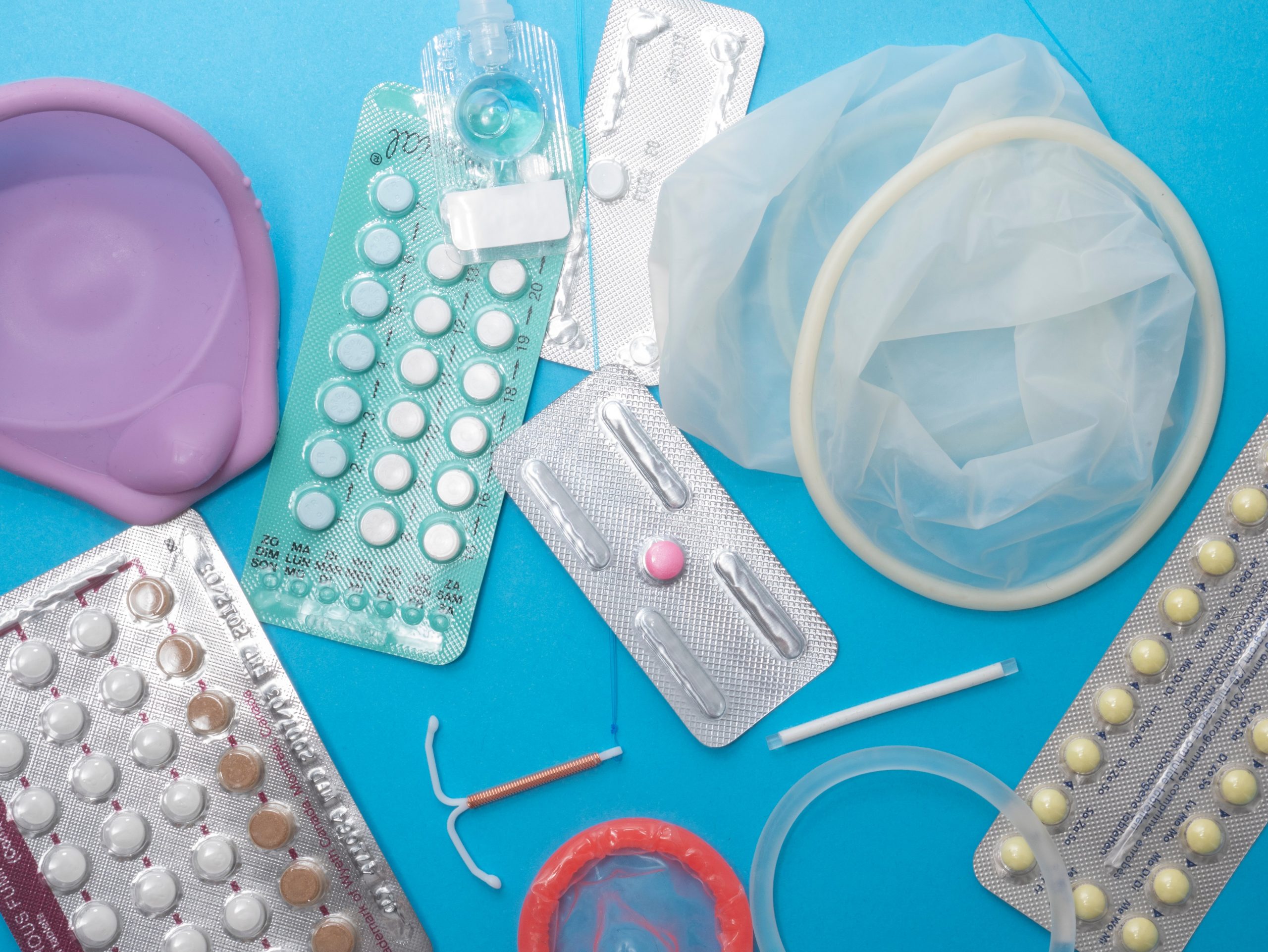
Finding the right contraception method shouldn’t be trial and error, says Elena Rueda Carrasco. She tells FemTech World why understanding contraception is more important than ever.
“Despite encouraging research, a male birth control pill remains elusive,” reads a recent headline in The New York Times.
Although, according to the article, scientists have been researching ways to create a male birth control pill since the 1970s, negative side effects, such as weight gain, acne, irritability and mood swings, have been responsible for not licensing any products for use.
“A woman can struggle with the same symptoms in silence – something rather normalised in our society,” says Elena Rueda Carrasco, medical scientist and co-founder and CEO of Dama Health, a company that offers personalised contraception recommendation. “Many women are being told from a very young age that the pain they are experiencing is normal,” she adds.
Indeed birth control side effects, such as weight gain, mood changes and headaches, affect eight in ten women, but are yet deemed too dangerous for developing a pill for men.
“The side effects and the impact of different contraception methods on women have been very normalised when you compare it to other areas of medicine,” Rueda explains. “In a lot of cases, women end up with conditions like endometriosis and PCOS – polycystic ovary syndrome – because their symptoms are often ignored and early diagnosis is not achieved.”

Elena Rueda Carrasco, co-founder and CEO of Dama Health
Figures suggest that medical consultations are too short to offer the right guidance and on average, women try 3.5 different contraceptive methods before finding the one that works for them.
“In the UK, we have such a fragmented [health] system,” the co-founder says. “As a woman, you can get your contraception through your GP, you can go to a sexual health clinic, you can speak to a nurse or a pharmacist. And from our own research, we found out that for many women, this is very confusing.
“Most of the time women will probably call or visit their GP and the GP is actually not a specialist,” Rueda points out. “The GPs are doing a bit of everything, with just a couple of contraception methods they’re comfortable prescribing. Therefore, communication is really hard when you have this fragmentation happening and you don’t really know where to go and who to speak to”.
Dama Health aims to personalise and tailor the way women are being prescribed contraception, screening for and identifying the side effects that women might experience individually. An algorithm would then match them to the recommended contraceptive options that are most suitable for them.
“As a team, we are all scientists and doctors by background, and we were all experiencing this problem of trial and error in the way that women were being prescribed hormonal medication and in this specific case, contraception,” the CEO says.
“I think it all came from personal experience and we all felt that it was an issue. Our chief medical officer, Dr Aaron Lazorwitz, is an OBGYN doctor and he experienced it in clinic, whereas myself and Paulina Cecula [her co-founder] experienced it as women. Paulina is also a medic and she was seeing that actually, doctors were having difficulties and the whole thing ended up being like ‘try this and come back if there’s a problem’.
“We thought that there’s a huge inefficiency [within the system] in this day and age. Women shouldn’t be having to trial and error to find if something is good for them,” Rueda adds. “So that drove us to start doing screening tests, making everything a lot more personal and helping the medical community to make the best decisions when it comes to contraception methods.”
She says that at the heart of this, education and communication are extremely important. “Women and young adults need to be educated around the topic of contraception better,” the CEO says.
“Women especially feel unable to verbalise how they feel, they can’t actually communicate it. So part of our solutions through the screening process, is to ask the right questions that help women understand their bodies and have a better conversation with their doctor.
“For that reason, I think personalised medicine is the future because actually, this one-size-fits-all approach isn’t working. When it comes to medication, you really do need to get that personalisation because the inside of us is so different and so unique.
“With technology becoming cheaper, we have the tools and the ability to make it more personalised and that essentially means taking into account medical history, preferences, maybe some biomarker information and just putting them together to be able to tailor the right treatment option.”
Experts like Rueda believe that very often huge amounts of data are being lost because of a lack of a data tracking system and that subsequently, makes the entire research process harder. “A doctor doesn’t tell you ‘Hey, try this contraception and tell me how you feel’ and actually write a report on it,” she explains. “That information is, therefore, not being captured. That’s dangerous because so much information is not being taken into account.”
But how could data tracking help the medical world? “A doctor is always going to use quantitative and qualitative data to make decisions, so symptom-tracking is one of the most powerful things that we can do as patients,” Rueda adds. “Data has so much power because it helps you essentially prove your point in an argument and is the fastest way to get information and to accelerate research.”
Walking into the clinic, as a woman, and feeling empowered is something that the CEO would like to see in the future and she also hopes that her company will support all women equally.
“The most important thing [for Dama Health] is to be able to be as accessible as possible and help women from different backgrounds. That impact is really important for us and also just working with the medical community and trying to implement our screening tests that could be easily accessed by anyone.”
The team will also be recruiting for beta testers and clinical in Q4 of 2022. If you are UK-based, you can sign-up here to be part of future clinical trials and research programmes.
For more info, visit damahealth.com.
News
Opinion: Women don’t need a refreshed health strategy – we need action

By Justyna Strzeszynska, founder of menstrual health platform Joii
The Government’s announcement that it will renew the Women’s Health Strategy is, on the surface, good news.
The original strategy in 2022 was historic – the first time women’s health had been acknowledged as something that required its own plan.
It raised awareness, started conversations and encouraged women to come forward and talk about their health.
But awareness alone hasn’t changed much on the ground.
Women are still waiting years for diagnoses, gynaecology waiting lists are still some of the longest in the NHS and many women are still being told their symptoms are ‘just part of being a woman’, especially when it comes to periods, pain or fatigue.
If the Government is going to refresh this strategy, we need to be honest about what didn’t work last time and what has to change now.
One issue with the previous strategy was the way it focused on specific conditions.
Endometriosis and PCOS were rightly brought forward and the advocacy behind that has been extraordinary. But women’s health can’t work like a spotlight, where each year a new condition is added based on who campaigns most effectively.
Some of the most common and life-disrupting conditions still sit in the background.
Heavy menstrual bleeding affects one in three women. Fibroids affect up to one in three by age 50. Adenomyosis is thought to affect one in ten.
These aren’t rare conditions, they are everyday realities. Yet they receive less attention, less funding and far fewer structured care pathways.
They also disproportionately affect Black women, who are more likely to have severe symptoms and less likely to be believed.
If a renewed Women’s Health Strategy is going to address inequality, then these conditions can’t remain an afterthought.
The other major issue is how diagnosis actually happens.
Right now, if you go to your GP with heavy bleeding or pelvic pain, the first questions are usually ‘how much blood do you think you’re losing?’ and ‘how bad is the pain, on a scale of 1 to 10?’
Most women have never been taught what ‘normal’ bleeding looks like and their pain has become background noise. Many also feel unsure or embarrassed about describing symptoms accurately.
So women hesitate, clinicians hesitate and referrals get delayed. That’s how we end up with eight-year diagnostic journeys.
If we want to reduce waiting lists and speed up diagnosis, we need to fix the front door.
First, we need to give GPs standardised tools to measure menstrual bleeding and symptom impact.
One of the biggest barriers to diagnosing menstrual health conditions is that we still rely on women to estimate their bleeding and pain with no reference points.
Most women, and especially young girls, don’t know what counts as heavy bleeding and many have normalised symptoms that could actually be clinical red flags.
Without standard measurement, clinicians can’t triage effectively and women fall into long cycles of ‘wait and see’.
The renewed strategy should introduce validated digital and clinical tools, so patients and clinicians are working from the same evidence, not guesswork.
Second, expand and standardise Women’s Health Hubs so access isn’t determined by postcode.
Women’s Health Hubs already exist in most of England, which is a strong start, but not all hubs offer the same services, capacity or quality of care.
Some are genuinely transformative while others function more as signposting centres.
To actually reduce the backlog and speed up diagnosis, hubs need to be properly resourced and consistent, with clear referral pathways from primary care.
The refreshed strategy should set national standards for what every hub must deliver so accessing timely assessment isn’t dependent on where a woman happens to live.
Finally, there needs to be a shift towards treating menstrual and pelvic conditions as chronic, not occasional episodes.
Conditions like endometriosis, adenomyosis, fibroids, PCOS and chronic pelvic pain don’t follow single-appointment cycles yet our system is structured as if they do.
Women are often seen once, reassured and discharged, only to start the entire referral process again when symptoms worsen. This wastes NHS time and leaves women feeling unheard.
The renewed strategy needs to support ongoing monitoring and follow-up, recognising these conditions as long-term health issues requiring continuous management, not episodic care.
Most importantly, the refreshed strategy must come with clear timelines, ringfenced funding and actual accountability.
Otherwise, we end up with another web page and a press release, instead of change.
Women are already doing their part by speaking up.
Now the system needs to meet them.
Wellness
AI-powered women’s health companion Nexus launches in UK

The first AI-powered health companion designed exclusively for women is launching in the UK today.
Nexus provides a unified fitness, nutrition, medication and cycle tracking solution alongside a personal digital health coach.
Unlike other health apps built for isolated concerns, Nexus recognises that women’s health is interconnected.
Powered by a proprietary AI model and guided by Nova, the in-app AI health coach, Nexus connects the dots between every aspect of a woman’s wellbeing, from hormonal cycles to nutrition and mental health, and provides personalised, evidence-based guidance in real time.
Co-founder Leo Tyson has worked with over a thousand women in his role as a personal health coach, but wanted to support even more women through their health journeys.
Tyson said: “Nexus gives every woman a health coach in her pocket and the knowledge to become her own expert.
“I would see women desperate for guidance but unable to afford one-on-one coaching.
“They would try to patch together information from different apps or cheaper coaches, often making things worse rather than better.
“At the same time, the sector has been missing an integrated platform that understands women’s health is not just their cycles, but their whole health story.
“Our mission with Nexus is to give every woman at every life stage the personalisation, clarity and support of a one-to-one health coaching, at an accessible price.”
The launch draws on extensive research in Nexus’ white paper The Period App Problem, which revealed that many women feel disappointed by menstrual tracking apps that fail to deliver meaningful, personalised insights.
Nexus bridges this gap with a female-specific onboarding process offering over 50,000 unique combinations of personalised wellness insights.
With Nova, users also have access to an AI coach that truly knows them, offering adaptive, actionable guidance grounded in science and tailored to each woman’s unique physiology and life stage.
The vision behind Nexus is to give women control over their own health data, using it to improve conversations with healthcare providers, reduce medical gaslighting and accelerate diagnosis times through advocacy and education.
At the heart of Nexus lies a proprietary large language model (LLM) and peer-reviewed health database, built specifically for women.
This architecture blends medical research, clinical guidelines and user data to generate precise recommendations, far surpassing the capabilities of off-the-shelf AI systems.
Nexus is available on the App Store from today (10th October).
Mental health
Black and Latinx women more likely to experience serious complications in planned repeat caesareans

Black and Latinx women are more likely to face serious complications during planned repeat caesareans than White women, new US research has found.
While complication rates were similar across all racial and ethnic groups for vaginal births after a previous caesarean, disparities emerged in outcomes for those having planned repeat surgery.
Researchers examined severe maternal morbidity – serious complications during childbirth that can have lasting health effects – across 72,836 births between 2012 and 2021 among people who had previously delivered by caesarean.
The team analysed data from the Massachusetts Pregnancy to Early Life Longitudinal Data System, focusing on births following an earlier caesarean.
The 21 complications tracked include outcomes such as hysterectomy (removal of the womb), heart attack, embolism (blood vessel blockage), kidney failure, eclampsia (seizures in pregnancy linked to high blood pressure), sepsis (a life-threatening infection), and complications related to anaesthesia.
Laura Attanasio is associate professor of health policy and management and lead author of the study.
She said: “There’s been increasing recognition in recent years that the US has this rising rate of severe maternal morbidity, which can have a negative impact on one’s health in the future.”
It also can be considered a near miss for maternal mortality, or death, which is also high in the US relative to other wealthy countries, though rare.
The study examined three birth scenarios: vaginal birth after caesarean (VBAC), planned repeat caesarean, and unplanned repeat caesarean – where someone intends to deliver vaginally but ultimately requires surgery.
Attanasio said: “Among White birthing people, severe maternal morbidity rates were similar for VBAC and for planned repeat caesarean.
“But for Black and Latinx birthing people, planned repeat caesarean had a higher rate of severe maternal morbidity compared to VBAC.”
Among all groups, the highest rate of complications occurred during unplanned repeat caesareans.
In this category, however, disparities between racial and ethnic groups were not observed.
The study population was 56.8 per cent White, 20.1 per cent Latinx, 11 per cent Black and 12.1 per cent who identified as another race or ethnicity.
The researchers used hospital discharge and birth records to identify medical issues and demographic data including race, ethnicity and parental birthplace.
They adjusted for medical risk factors more commonly associated with marginalised groups, suggesting other influences may be contributing to the differences in outcomes.
Attanasio hypothesised that “quality of clinical care can be worse for people from marginalised racial and ethnic groups, either because they’re being cared for in settings that are lower resourced and less able to provide quality care, or in some cases they could be receiving worse care in the same setting as White birthing people due to structural or interpersonal racism.”
The findings suggest the need to identify and address factors contributing to higher complication rates among Black and Latinx individuals during planned repeat caesareans.
“Future work should identify interventions to improve quality of care and promote equity for this population,” the researchers said.

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoThree sessions that show exactly where women’s health is heading in 2026

 Entrepreneur3 days ago
Entrepreneur3 days agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 Pregnancy4 weeks ago
Pregnancy4 weeks agoHow NIPT has evolved and what AI NIPT means in 2026

 News3 weeks ago
News3 weeks agoTwo weeks left to make your mark in women’s cardiovascular health

 Opinion4 weeks ago
Opinion4 weeks agoQ1 momentum: Female founders are advancing, but the system still hasn’t caught up

 Fertility2 weeks ago
Fertility2 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic

 Menopause2 weeks ago
Menopause2 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 News2 weeks ago
News2 weeks agoSelf-employment linked to better cardiovascular health outcomes in Hispanic women





























Pingback: Pill problems: Why are women still struggling to access the right contraception, and how can healthtech help? - FemTech World