Entrepreneur
Could the first instant at-home test transform how women track their hormones?

Eli Health is on a mission to transform female hormone testing with the first FDA-registered instant hormone monitoring system. Co-founder and CEO, Marina Pavlovic Rivas, speaks to Femtech World about making hormone testing more accessible.
The Hormometer, developed by femtech company, Eli Health, enables real-time, saliva testing for instant results, enabling women to monitor their hormonal fluctuations from the comfort of their own home.
Developed over five years, Eli Health raised US$12m in its Series A funding to scale the product, bringing its total funding to US$20m.
The FDA-registered Hormometer is an at-home needle-free test kit that utilises AI for data-driven insights. Currently, the Hormometer enables women to test their progesterone and cortisol levels, with estradiol testing currently in development.
While women have traditionally paid hundreds of pounds for tests, potentially waiting weeks for test results, Eli’s Hormometer uses computer vision algorithms to analyse the saliva samples, delivering results in 20 minutes at around UD$8 per test.
Marina Pavlovic Rivas, co-founder and CEO of Eli Health, which has 12 patented inventions for hormone monitoring, says that the tests and accompanying app offer insights into women’s health across fertility, menopause, and mental health, enabling improved access and affordability to hormone testing.
“We started the company because we wanted women to have access to the important data when it comes to their health,” Rivas tells Femtech World.
“We realised that when it comes to hormones, this data doesn’t exist at the frequency it’s needed, because you can test your hormones through a lab, but that process takes at best a few days, and on average, a few weeks. It is quite expensive as well.”
Traditional hormone lab tests only provide insight into a certain window of time, Rivas explains, but for women, whose hormones are in constant fluctuation, more consistent monitoring is needed. Hormones like cortisol can also fluctuate up to 100 per cent in one hour, making it difficult to get a full picture.
“This frequency and cost don’t enable high frequency, long-term testing, which reflects, ultimately, the fluctuations of hormones that happen on a daily basis,” Rivas says.
“Our mission became to create that real-time interface for the human body, so that we can have access to this data at the frequency that matters.”
Using lateral flow assays, the test works by collecting saliva from the tongue. The individual then uploads a picture of the test into the app.
The app utilises AI-driven insights to provide lab-grade results on hormone levels, along with scores, benchmarks and recommendations to enable women to take action.
“What we enable is not just cheaper, faster testing,” says Rivas. “It’s a very different approach to this type of data. Let’s say you’re measuring your heart rate at the doctor’s office once per year, it’s very different to having it on your wrist at all times with a smart watch. That becomes information that you can use on a daily basis to take actions around your lifestyle, for example, sleep, exercise, and all areas of your health and wellness.”
Explaining the range of markers measured by the Hormometre, she continues: “It touches the typical areas of women’s health – fertility and menopause, but also endocrine conditions and beyond that, some of our hormones, like cortisol, are not reproductive hormones. For us, it was important to include them, because when we speak of hormonal health, it goes beyond reproductive health.
“Some biomarkers like cortisol have a major impact on mental health, how people feel on a daily basis, how they perform at work, but also when they exercise and ultimately, all aspects of their health. It is our mission to enable people to improve their health and performance across all of those different areas.”
Rivas adds: “Around 80 per cent of women will experience symptoms related to hormonal imbalances in their lives, so that’s a very high number of people. When it comes to some hormones like cortisol, there’s one in three people who experience dysregulation.
“How that translates on a day-to-day basis is that people live with different symptoms that impact their work, that impact personal lives, and having that information enables them to manage those symptoms and even eliminate them.”
It is not only about monitoring hormones, as Rivas highlights, but also about taking preventative action to ward off potential chronic conditions in the future.
“It has been shown in research again and again that dysregulation of some hormones, like cortisol, is also linked to higher probability of developing different conditions, including chronic conditions, heart conditions, cognitive disease and more,” says Rivas.
“Tracking hormones has the double benefit of feeling better today, but also preventing different conditions tomorrow.”
Having recently won the Femtech World Brain and Mental Health Innovation award for its work to help women better understand the impact of hormones on their mental health, EliHealth is now focusing on expanding to other markers.
“We are always focused on making a product that can address the needs of our users, and we’re always very focused on that work. So, to be able to take a step back and have recognition from the industry is meaningful for us in our mission,” Rivas adds.
“The future will be about continuing to focus on that mission of making a real-time interface to the human body by expanding to other hormones and their markers, and ultimately providing that information in real time to our users.”
Eli Health won the Brain and Mental Health Innovation category at the Femtech World Awards 2025. See full winners list here.
Entrepreneur
Sun Pharma to acquire Organon in US$11bn deal
News
Women’s digital health market set to reach US$5.28 billion in 2026 – report
Entrepreneur
Future Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

Future Fertility Inc. has announced the closing of a US$4.1 million Series A financing round.
The round was led by M Ventures (the corporate venture capital arm of Merck KGaA, Darmstadt, Germany) and Whitecap Venture Partners, with participation from new investors Sandpiper Ventures, Gaingels, and Jolt VC.
The financing will accelerate Future Fertility’s commercial expansion into Asia-Pacific and support its entry into the United States, including planned FDA 510(k) clearance for additional products as part of a broader U.S. market entry strategy.
Proceeds will also advance the development of a broader AI platform, from egg assessment through to embryo transfer, designed to support clinicians, embryologists, and patients across the full IVF journey.
M Ventures and Whitecap have supported Future Fertility’s mission to translate AI innovation into meaningful clinical outcomes since the company’s earliest stages.
Oliver Hardick, investment director, M Ventures, said: “Future Fertility is addressing a critical unmet need in reproductive medicine with a differentiated AI platform grounded in clinical data and real-world workflow integration.
“We are excited to continue supporting the company and team because we believe its technology has the potential to improve decision-making for clinicians, bring greater clarity to patients, and help advance a more personalised standard of care in fertility treatment.”
Future Fertility’s AI platform addresses a long-standing gap in fertility care: historically, there has been no objective, clinically validated method for assessing egg quality (Gardner et al., 2025), despite it being one of the most important drivers of reproductive success.
The company’s suite of deep learning tools includes VIOLET™, MAGENTA™, and ROSE™, purpose-built for egg freezing, IVF, and egg donation respectively.
The tools are based on AI models trained and validated on more than 650,000 oocyte images and are deployed in over 300 clinics across 35 countries.
Rhiannon Davies, founding and managing partner, Sandpiper Ventures, said: “The best outcomes in fertility care globally come from better data and smarter tools. Future Fertility understands that, and they’ve built a platform that delivers on it.
“Sandpiper is proud to back a team turning rigorous science into real results for patients and clinicians alike.”
Partnerships with the world’s leading fertility networks – including IVI RMA and Eugin Group across Latin America and Europe, FertGroup Medicina Reproductiva in Brazil, and most recently announced Kato Ladies Clinic in Japan – reflect growing demand for objective, AI-powered oocyte assessment in fertility care. In the United States, ROSE™ is newly available under an FDA 513(g) determination.
Research shows that approximately 50 per cent of IVF patients do not understand their likelihood of success, and many discontinue treatment prematurely, even though cumulative success rates improve significantly with multiple cycles (McMahon et al., 2024).
By delivering earlier clarity on egg quality, Future Fertility’s tools support more informed conversations between clinicians and patients, helping set realistic expectations and guide decisions about next steps.
Future Fertility’s growing evidence base spans seven peer-reviewed publications in Human Reproduction, Reproductive BioMedicine Online, Fertility & Sterility, and Nature’s Scientific Reports, and more than 70 scientific abstracts accepted and presented with partner clinics at conferences worldwide.
Christine Prada, CEO, Future Fertility, said: “Fertility treatment is one of the most emotionally and physically demanding experiences a person can go through.
“Every patient deserves objective data, not just a best guess, to support better decisions at critical moments in their care.
“This funding means we can bring that clarity to more patients, in more countries, at a moment when it matters most.”
Find out more about Future Fertility at futurefertility.com

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
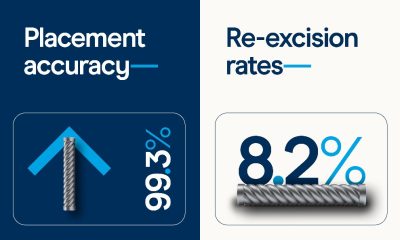
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Mental health4 weeks ago
Mental health4 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News4 weeks ago
News4 weeks agoResistance training has preventative effects in menopause, study finds

 Events2 weeks ago
Events2 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now






























1 Comment