Fertility
Artificial insemination kit granted FDA clearance
Mosie Baby becomes the first company to receive FDA clearance for at-home intravaginal insemination
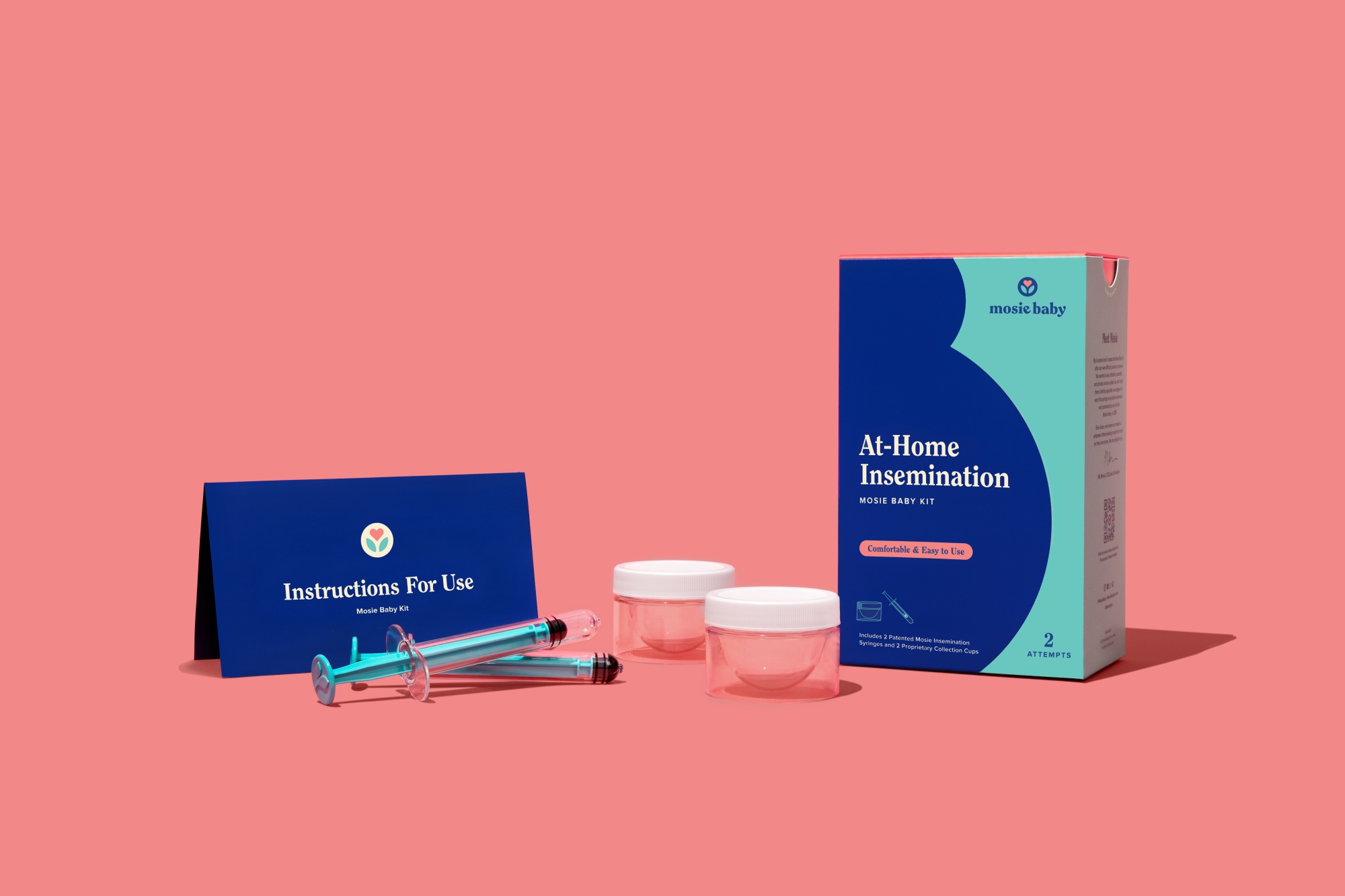
The US fertility start-up Mosie Baby has received FDA Class II clearance for its at-home insemination kit.
The kit, selected by the Bill and Melinda Gates Foundation to be part of the Designing Motherhood exhibition at the Discovery Center Museum in Seattle, aims to support those unable to conceive with intercourse or for whom intercourse is not an option.
Designed to be used with either a fresh or cryogenically frozen donor semen sample, each kit includes two syringes for at-home insemination and two collection cups for semen collection. The product is the first and only FDA-cleared over-the-counter kit for use in intravaginal insemination (IVI).
“Nearly 10 years ago, my husband and I were devastated by a diagnosis of unexplained infertility and were desperate for options that were safe, financially accessible and easy to use at home,” said co-founder and CEO Maureen Brown.
“Since inventing the Mosie Baby Kit in 2014, we realised we weren’t alone in our fertility journey as it is reported that one in six people experience infertility. To date, we’re very proud to share that Mosie Baby has helped more than 100,000 families inseminate from the comfort of their own home.
“We are now thrilled to offer our device as an FDA reviewed option for families looking to inseminate at home.”
Kwame Ulmer, managing partner at MedTech Impact Partners and former deputy director at FDA, said: “The recent Mosie Baby clearance means hundreds of thousands of underserved people now benefit from an at home option.
“The technology adheres to the highest FDA recognised test standards and is supported by robust clinical performance testing. The team took the time and care to manufacture a quality product.
“I am delighted to see it become accessible to countless future parents on their fertility journey. The team at Mosie Baby is on a path to becoming the at home gold standard fertility option.”
Following its FDA 510k Class II clearance, Mosie Baby is planning to expand access to its insemination kit, aiming to democratise access to family building and eliminate stigma associated with insemination.
The Mosie Baby insemination kit is available for purchase at mosiebaby.com, CVS.com or at select CVS stores nationwide. The kit is expected to launch with additional retailers and healthcare partners in 2024.
To receive the Femtech World newsletter, sign up here.
Fertility
Gum disease may impair female fertility and egg quality – study
Fertility
AI could transform ovarian care through personalisation, study finds
AI could transform ovarian care by personalising cancer and fertility treatment, but more clinical validation is needed before routine use.
A systematic review and meta-analysis found AI models showed high diagnostic accuracy for ovarian cancer when combining data such as ultrasound scans and blood test results.
Across 81 studies, AI models correctly identified ovarian cancer in around nine out of 10 cases, with pooled rates of 89 to 94 per cent.
They were also highly accurate at ruling out ovarian cancer when it was not present, with specificity of 85 to 91 per cent.
The analysis also found that explainable AI tools could predict complete surgical cytoreduction in advanced ovarian cancer.
Complete surgical cytoreduction means removing all visible cancer during surgery, which can be an important goal in treatment planning.
The tools achieved a pooled AUC of 0.87. AUC is a measure of how well a model distinguishes between different outcomes, with higher scores showing stronger performance.
In reproductive medicine, AI algorithms helped physicians optimise ovarian stimulation protocols and predict follicular growth during IVF.
Ovarian stimulation is the use of hormones to encourage the ovaries to produce eggs, while follicles are the small sacs in the ovaries where eggs develop.
The review found AI could reliably model ovarian response in IVF with a pooled AUC of 0.81.
However, researchers said challenges remain in translating promising research findings into routine clinical practice.
They identified substantial variation across studies, driven by retrospective study designs, variable AI systems and a lack of standardised validation.
Only 22 per cent of analysed studies reported prospective, multicentre external validation, where models are tested forward in time across multiple healthcare settings.
The authors called for rigorous validation to help close the gap between research and routine clinical practice, alongside standardised methodological and reporting frameworks, smooth integration with clinical workflow and robust governance to support responsible and ethical AI use.
They concluded: “Artificial intelligence is a transformative force in the management of ovarian conditions.
“In gynaecologic oncology, AI enhances every phase of care, from early detection and accurate diagnosis to prognostic stratification and surgical planning.”
In reproductive medicine, AI personalises ovarian stimulation and refines the diagnosis of heterogenous endocrine disorders such as PCOS.
PCOS, or polycystic ovary syndrome, is a hormonal condition that can affect periods, skin, weight and fertility.
Fertility
Housing, work and fertility stop Britons having the families they want – research

 Menopause7 days ago
Menopause7 days agoPerimenopause misinformation ‘putting women at risk’

 Opinion4 weeks ago
Opinion4 weeks agoWhat Maternal Mental Health Month reveals about where postpartum support actually breaks down

 News4 weeks ago
News4 weeks agoNIH Grant terminations disproportionately impact minority scientists, research finds

 Adolescent health4 weeks ago
Adolescent health4 weeks agoWUKA brings Period-Positive Pool Party to London Aquatics Centre to keep girls swimming through puberty

 Insight3 weeks ago
Insight3 weeks agoPCOS renamed after decade-long campaign to end ‘cyst’ misconception

 Events4 weeks ago
Events4 weeks agoWHIS 2026 unveils agenda and first speakers for the leading women’s health summit

 Mental health4 weeks ago
Mental health4 weeks agoCBT shows promise for menopause insomnia and hot flashes

 Insight4 weeks ago
Insight4 weeks agoOnline abuse and deepfakes ‘pushing women out of public life’




























1 Comment