News
Organon unveils study of medical device for postpartum haemorrhage control
Postpartum haemorrhage is a severe vaginal bleeding that can occur within a day of giving birth or up to 12 weeks postpartum

The women’s health company Organon has announced the first observational study of its medical device for postpartum haemorrhage control.
The company has said that the Obstetrics & Gynecology journal published the results of the RUBY study which reached its primary effectiveness outcome of treating abnormal postpartum uterine bleeding and postpartum haemorrhage (PPH) with Organon’s JADA System.
JADA is an intrauterine vacuum-induced haemorrhage control device intended to provide control and treatment of abnormal PPH, a severe vaginal bleeding after childbirth that can occur within a day of giving birth or up to 12 weeks postpartum.
“PPH is a potentially life-threatening obstetric emergency that can occur after childbirth and requires timely medical intervention,” explained lead author Dena Goffman, a maternal-foetal medicine subspecialist, and professor and vice chair for quality and patient safety in the Department of Obstetrics and Gynecology at Columbia University Irving Medical Center in New York.
“Appropriate management of abnormal postpartum uterine bleeding is critical to minimise the potential clinical consequences of PPH and its associated severe maternal morbidities, such as the need for blood transfusions, ICU admission or hysterectomy.
“The new study, which included a larger population than the pivotal trial, showed how the device is being used outside of a controlled clinical trial environment.
“The results indicate that the intrauterine vacuum-induced haemorrhage control device is an important tool for PPH management.”
The study assessed records of 800 patients across 16 US hospitals from October 2020 through March 2022 and evaluated JADA treatment of abnormal postpartum uterine bleeding and PPH in “real-world” settings.
Treatment success was defined as bleeding control after insertion with no treatment escalation or bleeding recurrence.
According to Organon, JADA achieved treatment success in both the vaginal (92.5 per cent) and cesarean birth (83.7 per cent) groups, regardless of the cause of the abnormal PPH.
When reviewing patient cases that received standard PPH interventions plus the JADA System, lower blood loss prior to device insertion was associated with lower severe maternal morbidity.
Patricia Carney, Organon director of medical affairs, said: “Severe maternal morbidity rates in the US are rising, and PPH is recognised as a major cause of some of these morbidities.
“While severe maternal morbidities occurred even at lower levels of blood loss in this study, higher rates of morbidity were associated with higher levels of blood loss prior to JADA treatment in conjunction with other interventions that may have been used.
“These data shed light on the importance of appropriate control of blood loss, and it’s encouraging that the RUBY study reinforced the JADA pivotal study results in a real-world setting.”
Organon said adverse events in RUBY were consistent with the study PEARLE and what would be expected when managing an obstetric emergency.
Three serious adverse device events were deemed possibly related to the device or procedure, all of which resolved with treatment, the company added.
Pregnancy
Early birth safer in high blood pressure pregnancies – study
News
Low insulin diet and avoiding four food groups may prevent menopause weight gain
News
Three brain and mental health innovators shortlisted for award

We are excited to reveal the three exceptional innovators shortlisted for the Brain & Mental Health Innovation Award at the Femtech World Awards 2026.
The award recognises groundbreaking work addressing the cognitive and emotional health challenges that uniquely and disproportionately affect women.
This award is sponsored by Women In Cloud, the global network of 120,000 women tech founders, executives, professionals and allies across 80 countries, united in their mission to make the tech ecosystem an inclusive force for change – and to unlock US$1 billion in new economic access by 2030.
The shortlisted entries will now be judged by a representative from Women In Cloud who will announce the winner at a virtual event on June 19.
Congratulations to the shortlist and thank you to everyone who entered or nominated.
Brain and Mental Health Innovation Shortlist

HealCycle is a clinical and social breakthrough targeting what founder Ananya Grover calls the “Silent Decade” – the years when women’s endocrine health and environmental stressors are routinely dismissed by traditional healthcare silos.
Under the clinical leadership of psychiatrist Dr. Aninda Sidhana, HealCycle monitors HPO-axis markers, addresses conditions like PMDD, and integrates an AI companion, Tara, built on the principle of Radical Empathy.
Backed by the WICCI National Women’s Mental Health Council and designed to meet WHO standards for gender-responsive care, HealCycle is replacing silence with science – from Delhi to the world.

Môr is reimagining cognitive wellbeing from the ground up with the first science-led, female-first nutritional system designed around how women’s brains actually work.
Grounded in compelling research – including findings from Weill Cornell Medicine showing accelerated metabolic brain decline during menopause transition – Môr’s patented AM/PM chrono-targeted architecture delivers the right ingredients at the right time: daytime mental clarity and stress resilience in the morning, nervous system recovery and sleep support in the evening.
With backing from Innovate UK and a clinical feasibility study in development, Môr is building for the invisible majority: cognitively depleted women who have been failed by a market that never built for them.

Véa is an AI-powered emotional operating system that builds a living cognitive profile of each user, helping women understand their thoughts, triggers and behavioural patterns through neuroscience-backed journalling.
Véa does not just record how women feel, it connects the dots across weeks, months and years to reveal the hidden architecture of their emotional world.
Having already analysed over 101,000 anonymised words journaled to identify 3,000+ specific cognitive distortions – such as ‘double-bind’ guilt and ‘catastrophising’ – Véa is closing the critical gender data gap in mental health.

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
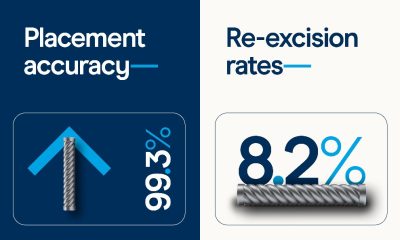
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy4 weeks ago
Pregnancy4 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now

 Wellness3 weeks ago
Wellness3 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Opinion2 weeks ago
Opinion2 weeks agoWhat Maternal Mental Health Month reveals about where postpartum support actually breaks down

 Fertility3 weeks ago
Fertility3 weeks agoToxins and climate harms having ‘alarming’ effect on fertility, research warns





























