News
Partnership to expand the clinical development of breast cancer drug candidate
ER+ breast cancers are estimated to account for 75-80 per cent of all breast cancer cases

The biotech company Lantern Pharma has announced a collaboration with TTC Oncology to expand the development of its breast cancer drug candidate.
Lantern Pharma has previously developed an artificial intelligence and machine learning platform to transform the cost, pace, and timeline of oncology drug discovery and development.
Now, through this latest collaboration, the company aims to use the insights from its AI platform to advance TTC Oncology’s first- and “best-in-class” drug candidate TTC-352 for recurrent ER+ breast cancer patients.
In US women, breast cancer remains the most commonly diagnosed cancer and second leading cause of cancer related deaths.
ER+ breast cancers are estimated to account for 75-80 per cent of all breast cancer cases and can have a recurrence rate between 13 per cent and 41 per cent.
Globally, the treatment of ER+ breast cancer is estimated to have a US$44bn market potential by 2027.
”It is of the utmost importance for cancer drug development to understand targeted tumour biology and mechanisms of resistance in order to select the patient population that will benefit the most from novel therapy,” said Dr Arkadiusz Dudek, TTC Oncology chief medical officer.
“We expect that by using Lantern’s RADR AI platform it can save us time and costs in the further successful clinical development of TTC-352 while providing important information for precision patient selection.”
The companies say the collaboration will be powered by the platform’s oncology-focused data points, algorithms and drug discovery and development modules and will be aimed at identifying biomarker or gene signatures to power potential patient selection for an upcoming clinical trial, characterising TTC-352’s mechanism of action and discovering additional treatment indications.
Panna Sharma, Lantern Pharma’s CEO and president, said: “Using AI insights generated by RADR, we are able to both sharpen existing clinical programmes and uncover additional unrealised clinical potential of Lantern’s and our collaborators’ drug candidates.
“We believe our AI-powered collaboration with TTC Oncology will accelerate the clinical development of TTC-352 for patients with metastatic ER+ breast cancer and will also identify new potential patient populations that can benefit from TTC-352 treatment.
Under the terms of the collaboration, Lantern Pharma is receiving an exclusive right to license TTC-352, including any collaboration intellectual property (IP), during an exclusive option period.
Additionally, Lantern and TTC will each participate in upfront, milestone, and royalty payments in the event a third party licenses IP resulting from the collaboration. No further financial details were disclosed.
Mental health
Poor sleep linked to Alzheimer’s risk in older women – study

Poor sleep may signal higher Alzheimer’s risk in older women with greater genetic risk, a study suggests.
Older women who reported poorer sleep also showed greater memory difficulties and more Alzheimer’s-related brain changes, the study found.
That pattern appeared only in women with higher genetic risk, suggesting sleep complaints may be a stronger warning sign for some women than for others.
Researchers examined 69 women aged 65 years and older taking part in the Women Inflammation Tau Study, an ongoing project focused on ageing and Alzheimer’s disease risk.
Participants completed questionnaires about their sleep quality, underwent memory testing and received brain scans measuring tau. Tau is a protein that accumulates abnormally in Alzheimer’s disease.
The study found that poorer self-reported sleep was associated with worse visual memory performance and greater tau accumulation in brain regions affected early in Alzheimer’s disease, but only among women with higher genetic risk.
Women with lower genetic risk did not show the same relationship between sleep complaints, memory and tau build-up. The finding was specific to visual memory and was not observed for verbal memory.
Researchers said the results add to growing evidence that sleep disturbances and Alzheimer’s disease may reinforce one another over time.
Previous studies have suggested that disrupted sleep can contribute to the build-up of abnormal tau proteins, while Alzheimer’s-related brain changes may also interfere with healthy sleep patterns.
Because women account for nearly two thirds of Alzheimer’s cases and frequently report poorer sleep quality than men, the researchers said sleep may represent an important and potentially modifiable risk factor in older women.
The authors noted that self-reported sleep assessments are inexpensive and easy to administer, raising the possibility that sleep complaints could help identify people who may benefit from closer monitoring or early intervention.
They also suggested that improving sleep could become a target for future Alzheimer’s prevention strategies, particularly for women at elevated genetic risk.
Fertility
AI could transform ovarian care through personalisation, study finds
AI could transform ovarian care by personalising cancer and fertility treatment, but more clinical validation is needed before routine use.
A systematic review and meta-analysis found AI models showed high diagnostic accuracy for ovarian cancer when combining data such as ultrasound scans and blood test results.
Across 81 studies, AI models correctly identified ovarian cancer in around nine out of 10 cases, with pooled rates of 89 to 94 per cent.
They were also highly accurate at ruling out ovarian cancer when it was not present, with specificity of 85 to 91 per cent.
The analysis also found that explainable AI tools could predict complete surgical cytoreduction in advanced ovarian cancer.
Complete surgical cytoreduction means removing all visible cancer during surgery, which can be an important goal in treatment planning.
The tools achieved a pooled AUC of 0.87. AUC is a measure of how well a model distinguishes between different outcomes, with higher scores showing stronger performance.
In reproductive medicine, AI algorithms helped physicians optimise ovarian stimulation protocols and predict follicular growth during IVF.
Ovarian stimulation is the use of hormones to encourage the ovaries to produce eggs, while follicles are the small sacs in the ovaries where eggs develop.
The review found AI could reliably model ovarian response in IVF with a pooled AUC of 0.81.
However, researchers said challenges remain in translating promising research findings into routine clinical practice.
They identified substantial variation across studies, driven by retrospective study designs, variable AI systems and a lack of standardised validation.
Only 22 per cent of analysed studies reported prospective, multicentre external validation, where models are tested forward in time across multiple healthcare settings.
The authors called for rigorous validation to help close the gap between research and routine clinical practice, alongside standardised methodological and reporting frameworks, smooth integration with clinical workflow and robust governance to support responsible and ethical AI use.
They concluded: “Artificial intelligence is a transformative force in the management of ovarian conditions.
“In gynaecologic oncology, AI enhances every phase of care, from early detection and accurate diagnosis to prognostic stratification and surgical planning.”
In reproductive medicine, AI personalises ovarian stimulation and refines the diagnosis of heterogenous endocrine disorders such as PCOS.
PCOS, or polycystic ovary syndrome, is a hormonal condition that can affect periods, skin, weight and fertility.
Cancer
Three cancer innovators shortlisted for Femtech World Award

Femtech World is delighted to reveal the shortlist for this year’s Women’s Cancer Innovation award.
The award, sponsored by Endomag, will honour a groundbreaking innovation dedicated to the prevention, early detection treatment or ongoing care of cancers that uniquely or disproportionately affect women.
Endomag is a medical technology company devoted to improving the global standard of cancer care.
Its Sentimag system, Magseed marker and Magtrace lymphatic tracer are used by thousands of the world’s leading physicians and cancer centres.
After careful review of this year’s submissions, we are delighted to announce the three shortlisted entries for the Women’s Cancer Innovation Award 2026.

Auria is tackling one of the most stubborn problems in breast cancer screening: the 66 per cent of women who simply don’t participate.
Rather than improving existing imaging pathways, Auria is creating an entirely new access layer: a non-invasive, at-home test that detects protein biomarkers for breast cancer in tears.
Auria’s test, a CLIA-certified Lab Developed Test, has been validated across more than 2,000 patients in multiple clinical studies with collaborators including MD Anderson Cancer Center and Stanford University.
It reports a sensitivity of 93 per cent and a negative predictive value of 98 per cent.

Founded on six years of combined research at the University of Barcelona and UC Irvine, The Blue Box has developed a non-invasive, urine-based test that detects breast cancer by analysing volatile organic compound (VOC) signatures – no radiation, no compression, no imaging facility required.
The test achieves a sensitivity of 88.42 per cent, outperforming mammography by 15 per cent overall, and by 30 per cent specifically in women with dense breasts.
The technology could function as a first-line screening tool in primary care settings, as a complement to mammography for high-density patients, or as an accessible alternative in healthcare systems where imaging infrastructure is limited.

Celbrea is a disposable and affordable thermal screening device that empowers women of all ages to stay on top of monitoring their breast health.
The device aims to add to doctors’ existing standard evaluation protocols with a quick, painless examination. Celbrea does not replace a mammogram but simply provides an additional way to screen for breast disease, including breast cancer.
The device consisting of two disposable pads with photochromic sensors. The pads are self-applied to each breast for 15 minutes.
1188 nano-sensors are embedded within a biocompatible multilayer pad, accurately measuring any temperature differences on the surface of the breast using liquid crystal thermographic technology.
What happens next
The shortlisted entries will now be judge by an Endomag representative who will reveal the winner at a virtual awards event on June 19.
Winners will receive a trophy and will be interviewed by a Femtech World journalist.

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
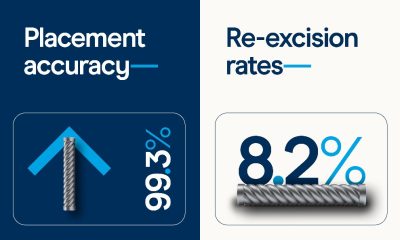
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy4 weeks ago
Pregnancy4 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Mental health4 weeks ago
Mental health4 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News4 weeks ago
News4 weeks agoResistance training has preventative effects in menopause, study finds

 Fertility3 weeks ago
Fertility3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now

 Wellness3 weeks ago
Wellness3 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage



























