To receive the Femtech World newsletter, sign up here.
News
Gender health gap study reveals ‘stark’ differences between men and women
The health differences between men and women grow with age, researchers have found

Women live longer than men but experience more years in poor health, a global gender health gap analysis has shown, underscoring the need for gender-responsive approaches to health.
The study, published in the Lancet Public Health journal, found that globally, there are substantial differences between men and women when it comes to health, with limited progress in bridging these health gaps over the past 30 years.
Non-fatal conditions that lead to health loss through illness and disability, including musculoskeletal conditions, mental health conditions and headache disorders, particularly affect women globally, the research suggested, while men are disproportionally affected by conditions that cause premature death, such as COVID-19, cardiovascular diseases and respiratory and liver diseases.
The health differences between men and women, however, continue to grow with age, leaving women with higher levels of illness and disability throughout their lives, as they tend to live longer than men.
“This report clearly shows that over the past 30 years global progress on health has been uneven,” said senior author, Dr Luisa Sorio Flor, at the Institute for Health Metrics and Evaluation (IHME), University of Washington.
“Females have longer lives but live more years in poor health, with limited progress made in reducing the burden of conditions leading to illness and disability, underscoring the urgent need for greater attention to non-fatal consequences that limit women’s physical and mental function.
“Similarly, males are experiencing a much higher and growing burden of disease with fatal consequences.
“This kind of critical, comparable and comprehensive research is important, both to understand the magnitude and distribution of the diverse and evolving health needs of females and males around the world and to identify key opportunities for health gain at all stages of life.”
The study is also a call for countries to boost their reporting of sex and gender data, said Sorio Flor.
“The timing is right for this study and call to action – not only because of where the evidence is now, but because COVID-19 has starkly reminded us that sex differences can profoundly impact health outcomes.
“One key point the study highlights is how females and males differ in many biological and social factors that fluctuate and, sometimes, accumulate over time, resulting in them experiencing health and disease differently at each stage of life and across world regions.
“The challenge now is to design, implement and evaluate sex- and gender-informed ways of preventing and treating the major causes of morbidity and premature mortality from an early age and across diverse populations.”
The study looked at the disparities in the 20 leading causes of illness and death between men and women, across ages and regions.
The modelling research used data from the Global Burden of Disease Study 2021, and did not include sex-specific health conditions, such as gynaecological conditions or prostate cancers.
The analysis estimated that for 13 out of the top 20 causes of illness and death, including COVID-19, road injuries and a range of heart, respiratory and liver diseases, the rate was higher in men than women in 2021.
Among the conditions evaluated, the study suggested the biggest contributors to health loss globally disadvantaging women are low back pain, depressive disorders, headache disorders, anxiety disorders, other musculoskeletal disorders, Alzheimer’s disease and other dementias, and HIV/AIDS.
These conditions predominantly contribute to illness and disability throughout life as opposed to leading to premature death, the research found.
For conditions with the greatest gap disadvantaging women, such as mental health conditions and musculoskeletal disorders, the differences in health loss between men and women begin early in life and continue to intensify with age, the findings also showed.
“Large causes of health loss in women, particularly musculoskeletal disorders and mental health conditions, have not received the attention that they deserve”, said co-lead author Gabriela Gil from IHME.
“It’s clear that women’s healthcare needs to extend well beyond areas that health systems and research funding have prioritized to date, such as sexual and reproductive concerns.”
She added: “Conditions that disproportionately impact females in all world regions, such as depressive disorders, are significantly underfunded compared with the massive burden they exert, with only a small proportion of government health expenditure globally earmarked for mental health conditions.
“Future health system planning must encompass the full spectrum of issues affecting females throughout their lives, especially given the higher level of disability they endure and the growing ratio of females to males in ageing populations.”
The global differences in health loss between men and women have been largely consistent for the past 30 years, but for some diseases, such as diabetes, the differences have grown, researchers found.
At the same time, there has been a disproportionate rise in global health loss caused by depressive disorders, anxiety, and some musculoskeletal disorders disadvantaging women.
The authors stressed that the health differences identified begin to emerge in adolescence, coinciding with a critical time when gender norms and attitudes intensify and puberty reshapes self-perceptions.
This pattern, they said, underscores the need for targeted responses from an early age to prevent the onset and exacerbation of health conditions and for adopting a life course approach when planning for health systems so that they are well-equipped to handle the needs of the populations they serve.
Dr Vedavati Patwardhan from the University of California, San Diego, said: “Our analysis highlights the need for targeted policies and planning to address the health needs of diverse populations.
“Without granular insights on risk behaviours, social dynamics, economic conditions and access to health care for all people in various parts of the world, the systemic barriers that sustain health inequities will remain.”
Fertility
AI could transform ovarian care through personalisation, study finds
AI could transform ovarian care by personalising cancer and fertility treatment, but more clinical validation is needed before routine use.
A systematic review and meta-analysis found AI models showed high diagnostic accuracy for ovarian cancer when combining data such as ultrasound scans and blood test results.
Across 81 studies, AI models correctly identified ovarian cancer in around nine out of 10 cases, with pooled rates of 89 to 94 per cent.
They were also highly accurate at ruling out ovarian cancer when it was not present, with specificity of 85 to 91 per cent.
The analysis also found that explainable AI tools could predict complete surgical cytoreduction in advanced ovarian cancer.
Complete surgical cytoreduction means removing all visible cancer during surgery, which can be an important goal in treatment planning.
The tools achieved a pooled AUC of 0.87. AUC is a measure of how well a model distinguishes between different outcomes, with higher scores showing stronger performance.
In reproductive medicine, AI algorithms helped physicians optimise ovarian stimulation protocols and predict follicular growth during IVF.
Ovarian stimulation is the use of hormones to encourage the ovaries to produce eggs, while follicles are the small sacs in the ovaries where eggs develop.
The review found AI could reliably model ovarian response in IVF with a pooled AUC of 0.81.
However, researchers said challenges remain in translating promising research findings into routine clinical practice.
They identified substantial variation across studies, driven by retrospective study designs, variable AI systems and a lack of standardised validation.
Only 22 per cent of analysed studies reported prospective, multicentre external validation, where models are tested forward in time across multiple healthcare settings.
The authors called for rigorous validation to help close the gap between research and routine clinical practice, alongside standardised methodological and reporting frameworks, smooth integration with clinical workflow and robust governance to support responsible and ethical AI use.
They concluded: “Artificial intelligence is a transformative force in the management of ovarian conditions.
“In gynaecologic oncology, AI enhances every phase of care, from early detection and accurate diagnosis to prognostic stratification and surgical planning.”
In reproductive medicine, AI personalises ovarian stimulation and refines the diagnosis of heterogenous endocrine disorders such as PCOS.
PCOS, or polycystic ovary syndrome, is a hormonal condition that can affect periods, skin, weight and fertility.
Cancer
Three cancer innovators shortlisted for Femtech World Award

Femtech World is delighted to reveal the shortlist for this year’s Women’s Cancer Innovation award.
The award, sponsored by Endomag, will honour a groundbreaking innovation dedicated to the prevention, early detection treatment or ongoing care of cancers that uniquely or disproportionately affect women.
Endomag is a medical technology company devoted to improving the global standard of cancer care.
Its Sentimag system, Magseed marker and Magtrace lymphatic tracer are used by thousands of the world’s leading physicians and cancer centres.
After careful review of this year’s submissions, we are delighted to announce the three shortlisted entries for the Women’s Cancer Innovation Award 2026.

Auria is tackling one of the most stubborn problems in breast cancer screening: the 66 per cent of women who simply don’t participate.
Rather than improving existing imaging pathways, Auria is creating an entirely new access layer: a non-invasive, at-home test that detects protein biomarkers for breast cancer in tears.
Auria’s test, a CLIA-certified Lab Developed Test, has been validated across more than 2,000 patients in multiple clinical studies with collaborators including MD Anderson Cancer Center and Stanford University.
It reports a sensitivity of 93 per cent and a negative predictive value of 98 per cent.

Founded on six years of combined research at the University of Barcelona and UC Irvine, The Blue Box has developed a non-invasive, urine-based test that detects breast cancer by analysing volatile organic compound (VOC) signatures – no radiation, no compression, no imaging facility required.
The test achieves a sensitivity of 88.42 per cent, outperforming mammography by 15 per cent overall, and by 30 per cent specifically in women with dense breasts.
The technology could function as a first-line screening tool in primary care settings, as a complement to mammography for high-density patients, or as an accessible alternative in healthcare systems where imaging infrastructure is limited.

Celbrea is a disposable and affordable thermal screening device that empowers women of all ages to stay on top of monitoring their breast health.
The device aims to add to doctors’ existing standard evaluation protocols with a quick, painless examination. Celbrea does not replace a mammogram but simply provides an additional way to screen for breast disease, including breast cancer.
The device consisting of two disposable pads with photochromic sensors. The pads are self-applied to each breast for 15 minutes.
1188 nano-sensors are embedded within a biocompatible multilayer pad, accurately measuring any temperature differences on the surface of the breast using liquid crystal thermographic technology.
What happens next
The shortlisted entries will now be judge by an Endomag representative who will reveal the winner at a virtual awards event on June 19.
Winners will receive a trophy and will be interviewed by a Femtech World journalist.
Insight
Common cancer marker may play active role in preventing the disease, study finds
Ki-67, a protein used to measure tumour growth, may also help prevent chromosome errors that drive cancer, a study suggests.
The findings could change how scientists view Ki-67, a marker commonly used in breast cancer and other tumours to assess how quickly cancer cells are growing.
Researchers found the protein may help preserve genome stability by maintaining the structural integrity of centromeres, key parts of chromosomes that help ensure DNA is shared correctly during cell division.
The research was led by professor Paola Vagnarelli at Brunel University of London in collaboration with scientists at the University of Edinburgh and the Technical University of Berlin.
Professor Vagnarelli said: “Doctors already measure Ki-67 to see how aggressive a cancer might be. But our results suggest it is actually helping maintain genome stability.
“That means it may be more than a marker. It could potentially also be a therapeutic target.”
The study examined three proteins that attach to chromosomes during cell division and help rebuild the molecular system that tells each new cell what kind of cell it is.
Every human cell carries identical DNA. What makes a liver cell different from a brain cell is which genes are switched on and which are kept inactive.
When a cell divides, that entire system of switches must be rebuilt. The three proteins involved in this process were Ki-67, Repo-Man and PNUTS.
Vagnarelli’s team developed a method that individually removes each protein from a living cell at the precise point of division. Older techniques could not isolate that moment cleanly.
They found that cells rely on all three proteins to reset themselves after division, but each failed in a different way when removed.
Without PNUTS, gene activity spiralled out of control and thousands of genes switched on at once.
Without Repo-Man, cells escaped safety checkpoints that usually stop damaged or abnormal cells from continuing to divide.
“What we didn’t expect was how clean the separation was,” said Vagnarelli.
Each protein fails in its own specific way. There is no redundancy, no safety net. Which means there are three separate points at which this process can go wrong.
“When the system breaks down, cells can emerge with the wrong number of chromosomes. That condition, called aneuploidy, is seen in disorders such as Down syndrome and in many cancers.
“We also found that these chromosome errors can trigger inflammatory signals inside the cell.”
Aneuploidy means a cell has too many or too few chromosomes, which can disrupt normal growth and function.
Inflammatory signals are chemical messages that can make a cell behave as if it is responding to injury or infection.
“These cells behave almost as if they are under attack,” said Vagnarelli.
“The immune response switches on because the genome is unstable.
“That link between chromosome imbalance and inflammation could help explain patterns we see in several diseases.”
The researchers said the findings may help cancer scientists better understand how chromosome instability, loss of gene regulation and cells dividing before they are ready contribute to tumour growth.
They said understanding the normal machinery that prevents these errors may help researchers find ways to push cancer cells into making mistakes they cannot survive.
“We now have a clearer map of the machinery that resets the cell after division,” said Vagnarelli.
“That knowledge gives us a starting point for thinking about new therapeutic approaches.”

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
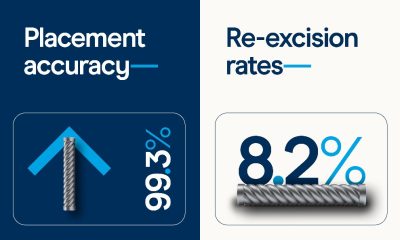
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Mental health4 weeks ago
Mental health4 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Wellness4 weeks ago
Wellness4 weeks agoResistance training has preventative effects in menopause, study finds

 Events2 weeks ago
Events2 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now


























3 Comments