Mental health
Women over 40 seeking raves for mental health benefits

Fertility
Housing, work and fertility stop Britons having the families they want – research
Menopause
CBT shows promise for menopause insomnia and hot flashes

Cognitive behavioural therapy (CBT) may offer short-term relief for menopause insomnia and night-time hot flushes, a pilot study suggests.
CBT is a structured, short-term talking treatment that helps people change thoughts and behaviours that can worsen sleep problems.
Researchers found the intervention was linked to meaningful short-term improvements in insomnia severity, hot flush interference, sleep self-efficacy, or confidence around sleep, and depressive symptoms.
The Menopause Society said insomnia affects an estimated 20 to 60 per cent of perimenopausal and postmenopausal women in the US.
Ongoing research is focusing on effective treatments because insomnia can have serious physical and psychological effects.
Dr Monica Christmas, associate medical director for The Menopause Society, said: “Nocturnal hot flushes (night sweats) and sleep disruption can have a significant effect on the quality of life with many women claiming extreme impairment due to symptoms that often start in early perimenopause and last 10 or more years.”
“Sleep disturbances can persist even in those using pharmacological therapy to manage hot flushes.
“The study’s findings highlight the utility of cognitive-behavioural therapy as a standalone treatment for insomnia and hot flushes, offering women an alternative or adjunct to pharmacological treatments.”
Insomnia is defined as disturbed sleep associated with distress or impaired daily functioning and is one of the most common complaints in perimenopause and postmenopause.
It can reduce quality of life and is linked to higher healthcare use and costs, disability, depression and cardiovascular disease.
Hot flushes occur in 60 to 80 per cent of women during the menopause transition and can persist for four to five years on average.
Night-time hot flushes are linked to sleep disruption, and women may respond by napping or spending longer in bed, which can help keep insomnia going.
Previous studies have shown that cognitive behavioural therapy is an effective treatment for insomnia and may also help women cope with hot flushes and other menopause symptoms.
However, few trials have looked at both insomnia and hot flushes together.
Insomnia during and after the menopause transition is complex and can have many causes, including ageing, hormone fluctuation, hot flushes, other sleep disorders, psychiatric and medical conditions and psychosocial stressors.
Because women with acute and sustained insomnia can experience greater negative health effects, effective treatment is important.
The pilot study concluded that CBT was feasible and may be a promising approach for menopause-related insomnia and nocturnal hot flushes, although the benefits appeared to lessen after three months.
Insight
Finding each other: Peer recognition as a clinical intervention in chronic illness

By Morgan Rose, chief science officer, Ema and Erlyn Macarayan, PhD, vice president, data science at PatientsLikeMe
May is Mental Health Awareness Month.
Most of the conversation this month treats depression and anxiety as standalone conditions, things people experience independently of their physical health.
For the patients we serve through the PLM platform, that framing leaves out something important.
Mental health in chronic illness functions as an overlay on a condition that does not end. It runs alongside the disease for as long as the disease lasts.
We have been analysing anonymised, aggregated conversational data from Ema, our agentic AI for patient support, and PLM users.
One pattern stands out across the dataset: what people are reaching for when they reach out.
The single most common behavior in the data, appearing in nearly 500 unique conversations, is the search for someone with the same diagnosis.
“Can you connect me with an MS group?” “Are there other people here with fibromyalgia?” “Does anyone in my city have what I have?” “I am looking for people like me.”
That request has a name in the research literature. It is peer support, and in the context of chronic illness, it functions as a mental health intervention.
The clinical case for peer recognition
The literature on peer support in chronic illness is well-developed.
Connection with others who share your diagnosis is associated with reduced depression severity, better treatment adherence, and a measurable drop in perceived isolation.
The mechanism behind those outcomes is recognition.
Someone else has lived inside the same symptom, navigated the same medication side effect, sat with the same diagnostic delay, and that recognition closes a specific gap that conventional therapy alone often cannot reach.
Therapists matter.
So do the people who know what an MS fatigue day actually feels like, what a fibro flare does to a person’s sense of self, what it is to be 34 and on a chemo regimen your friends cannot picture.
In clinical terms, that community is part of the care infrastructure for chronic illness, alongside medications, specialists, and labs.
PLM was built around this insight.
What Ema adds is a conversational layer that can route someone toward that community at the moment of need, before they have finished learning a platform.
“The PatientsLikeMe community has made living with MS manageable and in some bizarre way, even enjoyable sometimes because I’ve garnered these friendships and I am no longer afraid because all these other people are doing it with me.”
- PLM member living with MS
Why the burden is hard to address inside a clinical visit
There is a structural reason the mental health weight of chronic illness routinely goes undertreated. The visit is consumed by the physical condition.
The provider’s task list is long, the slot is short, and there is rarely a person in the room whose role is to ask how the patient is actually doing.
Some of that weight is also biological.
Depression in MS, for example, is roughly twice as common as in the general population, and is frequently undiagnosed because fatigue, cognitive change, and social withdrawal can be read as MS symptoms.
Similar overlap exists across cancer, autoimmune disease, and chronic pain.
Two systems run in parallel, shaped in part by the same underlying biology, yet routinely treated as separate.
That gap is where unguarded conversation tends to appear, and where the PLM data gets revealing.
The disclosure pattern
When mental health is mentioned in the PLM dataset, it rarely appears at the start of a conversation.
It surfaces sideways, after trust has been established by a clinical or logistical question.
One thread opens with questions about gabapentin and how PatientsLikeMe works.
A few exchanges later, the same user asks whether Ema has crisis resources. The conversation moves to feeling anxious, then depressed, then “I don’t know how I feel.”
Another thread spends several turns on MS management, medication questions, and which groups exist on PLM. Then the user writes, “My MS is making me feel overwhelmed and like everything is just too much. I’m not sure how to go on.”
Ema’s response in moments like that one matters.
She receives what was actually said, validates its weight, offers concrete steps for support, and connects the user back to the PLM community for the kind of isolation a clinical encounter cannot address.
The conversation pivoted because the user needed it to, and Ema followed.
That arc, the one that begins with a logistical question and ends in a disclosure about feeling unable to go on, is one of the clearest pictures we have of what the untreated chronic illness mental health burden looks like from the inside.
Crisis in the middle of an ordinary conversation
In 25 separate conversations in the PLM dataset, the mental health weight rose to the level of crisis. Users disclosed suicidal ideation directly.
One wrote, “I’m thinking of suicide.” Another asked what to do “if having a crisis and feeling suicidal.”
These conversations were happening on a patient platform, amid otherwise routine exchanges about a chronic condition.
The disclosures came in mid-thread, with no triage process to queue them.
Ema’s response was immediate and grounded. Hotline numbers, emergency services, an acknowledgment of the seriousness, and a reminder that the person is not alone.
The infrastructure to capture a moment like that at any hour, with no wait time, is something the conventional care system struggles to provide at scale. People are reaching for something in those moments.
Ema is built to be the thing they reach toward, and to hand them off to the human resources they need next.
What the data points toward
Pulling the patterns together, a coherent picture emerges.
People living with chronic illness carry a real and persistent mental health burden, and the burden tends to surface in the same conversations where they are managing medications, asking about treatment, and looking for others who share their diagnosis.
The most common request across the dataset is the request for community.
For PLM, that is the platform’s foundational thesis turning up in every dataset.
The platform was built on the premise that finding others with your condition is therapeutic. The conversation data confirms this, with patients explicitly asking for the connection.
For Ema, the implication is a design constraint.
We need to recognise when a question about gabapentin is an entry point into a question about feeling overwhelmed.
The route to peer recognition has to be as accessible as the route to clinical information.
And a moment of disclosure, whenever it arrives, has to land somewhere it can be received and responded to with care.
For Mental Health Awareness Month, the implication for chronic illness patients is direct.
Mental health in this population does not require a separate appointment that most patients will not make. It requires the people who already share the diagnosis to be part of the conversation, and it requires the platform to make that connection fast.
That is the work. It is what the data is asking us to build.
About Morgan Rose
Morgan Rose is chief science officer at Ema, an AI platform for patient health engagement.
Ema partners with health platforms and life sciences organisations to deliver clinically grounded, emotionally intelligent AI support where patients already are.
Learn more about Ema at emahealth.ai

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
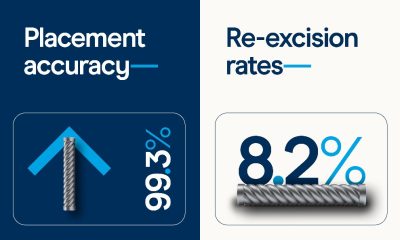
 Cancer3 weeks ago
Cancer3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both





























