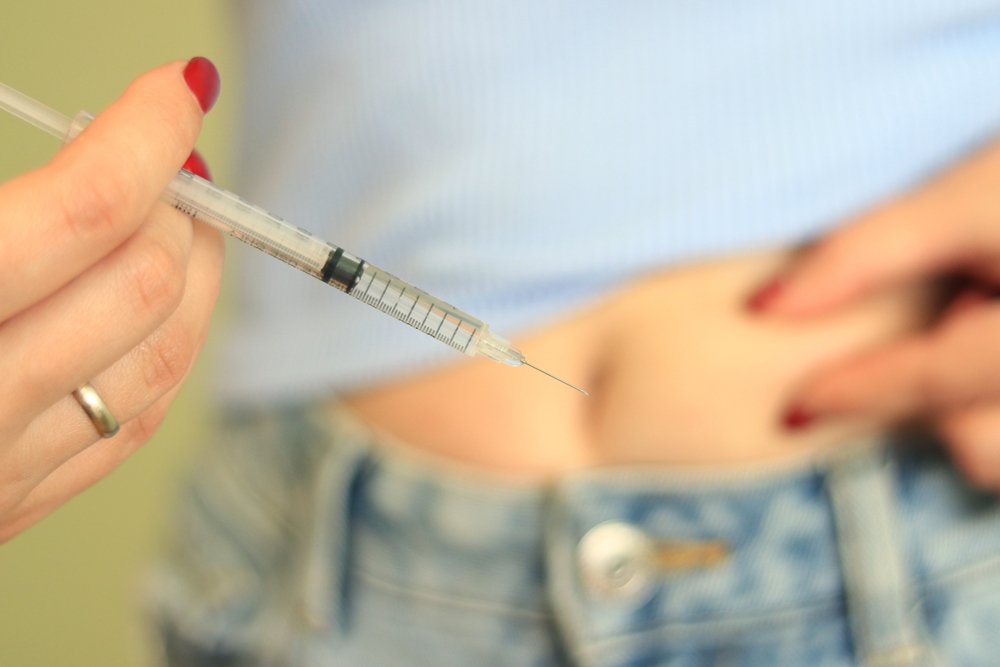
Fertility
IVF success rates higher if egg collection is done in the summer, say researchers
New study suggests the best conditions for live births appear to be associated with summer

The time of year when eggs are collected from women’s ovaries during fertility treatment could make a difference to live birth rates, new research has found.
Australian researchers have found that transferring frozen then thawed embryos to women’s wombs from eggs collected in the summer resulted in a 30 per cent higher likelihood of babies born alive than if the eggs had been retrieved in the autumn.
The team found a 28 per cent increase in the chances of a live birth among women who had eggs collected during days that had the most sunshine compared to days with the least sunshine.
“Over the duration of our study, the average live birth rate following frozen embryo transfer in Australia was 27 births per 100 people. In our study, the overall live birth rate following frozen embryo transfer was 28 births per 100 people,” said Dr Sebastian Leathersich, an obstetrician, gynaecologist and fellow in reproductive endocrinology and infertility at Fertility Specialists of Western Australia, City Fertility Centre, and the King Edward Memorial Hospital in Perth, Australia, who led the study.
“If eggs were collected in autumn, it was 26 births per 100 people, but if they were collected in summer there were 31 births per 100 people. This improvement in birth rates was seen regardless of when the embryos were finally transferred to the women’s wombs.
“The live birth rates when eggs were collected in spring or winter lay between these two figures, and the differences were not statistically significant.”
Until now, there have been conflicting findings on the effects of the seasons on pregnancies and live birth rates following egg collection and embryo freezing.
“It’s long been known that there is seasonal variation in natural birth rates around the world, but many factors could contribute to this including environmental, behavioural and sociological factors,” Leathersich explained.
“Most studies looking at IVF success rates have looked at fresh embryo transfers, where the embryo is put back within a week of the egg being collected. This makes it impossible to separate the potential impacts of environmental factors, such as season and hours of sunshine, on egg development and on embryo implantation and early pregnancy development.
“These days, many embryos are ‘frozen’ and then transferred at a later date. We realised this gave us an opportunity to explore the impact of environment on egg development and on early pregnancy separately by analysing the conditions at the time of egg collection independently from the conditions at the time of embryo transfer.”
Leathersich and his colleagues analysed outcomes from all frozen embryo transfers carried out at a single clinic in Perth over a period of eight years, from January 2013 to December 2021.
During this time there were 3,659 frozen embryo transfers with embryos generated from 2,155 IVF cycles in 1,835 patients. Information on outcomes was missing for two frozen embryo transfers and so these were excluded, leaving 3,657 for analysis.
The researchers looked at birth outcomes according to season, temperatures, and the actual number of hours of bright sunshine, as opposed to calculating hours from sunrise to sunset. They obtained the data on weather from the Australian Bureau of Meteorology. They created three groups for duration of sunshine on days when eggs were collected: low sunshine days, medium sunshine days and high sunshine days.
“When we looked specifically at the duration of sunshine around the time the eggs were collected, we saw a similar increase to that seen for egg collection during the summer,” Leathersich explained.
“The live birth rate following a frozen embryo transfer from an egg that was collected on a day with fewer hours of sunshine was 25.8 per cent.
“This increased to 30.4 per cent when the embryo came from an egg that was collected on days with the most hours of sunshine. When we took into account the season and conditions on the day of the embryo transfer, this improvement was still seen.”
The temperature on the day of egg collection did not affect the chances of a live birth. However, the chances of a live birth rate decreased by 18 per cent when the embryos were transferred on the hottest days compared to the coolest days and there was a small increase in miscarriage rates.
The study suggests that the best conditions for live births appear to be associated with summer and increased sunshine hours on the day of egg retrieval, Leathersich said.
“There are many factors that influence fertility treatment success, age being among the most important. However, this study adds further weight to the importance of environmental factors and their influence on egg quality and embryonic development,” he added.
“We effectively separated the conditions at the time of egg collection from the conditions at the time of transfer, demonstrating that environmental factors when the eggs are developing are as, if not more, important than environmental factors during implantation and early pregnancy.
“Optimising factors such as avoiding smoking, alcohol and other toxins and maintaining healthy activity levels and weight should be paramount. However, clinicians and patients could also consider external factors such as environmental conditions.”
Factors that may play a role in the increased live birth rates after egg collection in the summer and during more sunshine hours include melatonin. Levels of this hormone are usually higher in winter and spring, and eggs take three to six months to develop before they are released from the ovaries.
Differences in lifestyles between winter and summer months may also play a role, scientists say. The finding that miscarriage rates were highest when embryo transfer took place on the hottest days are consistent with epidemiological studies that show higher rates of miscarriage in the summer months.
“This suggests that the negative effects of high temperature are more likely related to early pregnancy rather than egg development,” said Dr Leathersich.
Limitations of the study include the fact that it is a retrospective rather than prospective study: looking back at what had already happened. For this reason, it can’t show that conditions at the time of egg collection cause the difference in live birth rates, only that they are associated with them.
“Ideally, these findings should be replicated in other sites with different conditions and different treatment protocols to confirm the findings,” Leathersich concluded.
“It would also be interesting to look at the impact of season and environmental factors on sperm parameters, as this could have contributed to our observations. We are now planning to analyse this same group of patients using air quality data, as there may be seasonal changes in exposure to harmful pollutants which could negatively affect reproductive outcomes.
“Finally, given the huge increase in so-called ‘social egg freezing’ for fertility preservation and the fact that this group generally have flexibility about when they choose to undergo treatment, it would be very interesting to see if these observations hold true with frozen eggs that are thawed and fertilised years later.
“Any improved outcomes in this group could have big impacts for women making decisions about their future fertility, but the long-term follow-up required means it is likely to be some time before we can draw any conclusions for this population.”
Insight
Why the UK’s fertility rate keeps falling – and what it means if you’re trying now

Article produced in association with Spital Clinic
The UK’s fertility rate has fallen for a third consecutive year to the lowest level ever recorded. That headline gets written every year, and it is easy to read it as a purely demographic story.
For anyone currently trying for a baby, the figure is something more practical: the conditions that produced the statistic are the same conditions shaping your own chances.
The decline has a clear pattern, and it is mostly not about couples being unable to conceive.
The change sits in when people start trying, and in what happens to fertility during the years by which most are now ready to have children.
What the numbers actually show
Figures from the ONS put the total fertility rate in England and Wales at 1.41 children per woman in 2024, down from 1.42 in 2023. The rate has been in overall decline since 2010 and has now recorded its lowest value three years running.
The figure sounds abstract until you compare it with the replacement level of 2.1 – the rate required for a population to sustain itself without net migration.
The UK has been below that line since the early 1970s, but the gap is now wider than at any point on record.
The data also shows where the decline is happening. Age-specific fertility rates for women in their twenties are the lowest of any generation since 1920. Rates for women in their thirties are holding up, and in some parts of the country rising.
Mothers are having babies later, not necessarily in smaller numbers. The average age of a first-time mother in England and Wales is now 31.0, up from 30.9 the year before. Regional variation matters too: London sits at 1.35, the West Midlands at 1.59.
Why the rate is falling
None of this is new. Every decade since the 1970s has seen the same trend, and it has accelerated in recent years. What has changed is the pace.
The shift is primarily social: delayed partnership formation, high housing costs, expensive childcare, and careers structured around full-time work through the exact years fertility is easiest.
The same pattern shows up across the EU, where the total fertility rate sat at 1.5 in 2022.
These forces compound. People meet later, partner later, feel financially ready later, and start trying later.
For many couples, first attempts happen in the early thirties, by which point fertility has begun its slow and uneven decline. A low national TFR is the population-level consequence of millions of individual timing decisions made under real-world constraints.
What this means for individuals trying now
Around one in seven couples in the UK will struggle to conceive naturally.
That figure has been stable for decades; the population of people seeking help, however, has grown – not because fertility itself has worsened, but because more people are trying during the window where it becomes harder.
UK fertility treatment data from the HFEA shows around 52,400 patients had over 77,500 IVF cycles in 2023, making 1 in every 32 UK births IVF-conceived.
The average age of a first-time IVF patient in the UK is now just over 35 – nearly six years older than the average first-time mother in the population overall.
NHS-funded IVF cycles have fallen from 40 per cent of the total in 2012 to 27 per cent in 2022, and to 24 per cent in England in 2023. The private sector has absorbed the rest.
When to get checked – and what it involves
Current NHS advice is to see a GP after a year of regular unprotected sex without a pregnancy, or sooner if you are 36 or older.
That threshold reflects the fact that every additional six months of trying is more clinically informative in the years when fertility is starting to shift.
The first set of investigations is usually straightforward.
For women, this typically covers hormone testing (AMH, FSH, LH, TSH and prolactin), rubella immunity, chlamydia screening, a mid-luteal progesterone and a transvaginal ultrasound.
For men, a semen analysis is the first step.
A private trying-to-conceive screening covers the same ground without the NHS waiting list, with the advantage that results can be reviewed in a single consultation.
The purpose of early screening is not to diagnose infertility – most couples conceive naturally within a year or two – but to identify specific, treatable issues before more time passes.
The fertility window is narrower than most people think
The uncomfortable truth behind the falling TFR is that the biological fertility window has not changed. The subtle decline begins around age 32, and accelerates from the late thirties.
The chance of natural conception in any given month is substantially lower at 40 than at 30, and falls sharply through the early forties.
IVF success rates track the same curve.
For patients aged 18 to 34, the average birth rate per embryo transferred was around 35 per cent in 2022; for those aged 40 to 42, around 10 per cent using their own eggs.
This is why the growth areas in UK fertility care are now pre-conception screening and elective egg freezing – HFEA data shows egg storage cycles rose from 4,700 in 2022 to 6,900 in 2023, one of the fastest-growing treatments in the sector.
A focused fertility consultation earlier in the timeline – in the late twenties or very early thirties, before there is a known problem – tends to produce better decisions than a consultation triggered by a year of trying without success.
The wider picture
The UK’s falling fertility rate is the product of a society that has reorganised when people have children, not one in which couples have become less capable of conceiving.
There is no need for alarm in that finding. The practical takeaway is that the old default of ‘wait and see’ assumes a timeline no longer matching the one most people now live.
For anyone currently trying, or planning to try soon, the single most useful move is to understand your own numbers earlier than previous generations did.
The national trend is not going to reverse quickly.
A clear picture of your own fertility window – and the information to use it well – is within reach in a way the headline statistics are not.
If you are trying to conceive or thinking about starting, a structured pre-conception review is a reasonable first step.
Disclaimer: This article is produced for informational purposes only and does not constitute medical advice, diagnosis or treatment. Clinical guidance referenced reflects published NHS, ONS and HFEA data as at April 2026. Individual circumstances vary; readers are advised to consult a qualified healthcare professional before acting on any information in this article. This piece was produced in association with Spital Clinic, which provided background clinical information for editorial purposes. Hyperlinks to external sources are included for reference only and do not represent an endorsement of any product, service or organisation.
Fertility
Toxins and climate harms having ‘alarming’ effect on fertility, research warns

Simultaneous exposure to toxic chemicals and climate-related heat may be worsening fertility harms across humans and wildlife, research suggests.
The review of scientific literature looks at how endocrine-disrupting chemicals, often found in plastic, together with climate-related effects such as heat stress, are each linked to lower fertility and fecundity, meaning the ability to reproduce, across species including humans, wildlife and invertebrates.
Though the reproductive harms of each issue in isolation are well studied, there is little research on what happens when living organisms are exposed to both.
“Together, the two issues are likely to pose a greater threat to fertility, and the additive effect is “alarming”, said Susanne Brander, a study lead author and courtesy faculty at Oregon State University.
“You’re not just getting exposed to one, but two, stressors at the same time that both may affect your fertility, and in turn the overall impact is going to be a bit worse,” Brander said.
The paper looked at 177 studies.
Shanna Swan, a co-author on the new paper, co-produced a 2017 study that found sperm levels among men in western countries had fallen by more than 50 per cent over four decades. Other research has suggested human fertility has been declining at a similar rate.
The University of Washington’s Institute for Health Metrics and Evaluation has previously said the world was approaching a “low-fertility future”, with more than three quarters of countries below replacement rate by 2050.
The new paper’s authors focused on the effects of endocrine-disrupting chemicals and substances, including microplastics, bisphenol, phthalates and PFAS.
These are thought to cause a range of serious reproductive problems, disrupt hormones and be a potential driver of falling fertility.
Brander said the harms linked to these chemicals are often similar across organisms, from invertebrates to humans.
Phthalates, for example, have been linked to altered sperm shape in invertebrates, spermatogenesis in rodents, meaning sperm production, and reduced sperm counts in humans.
PFAS are also thought to affect sperm quality, and both have been linked to hormone disruption.
The chemicals are widespread in consumer goods, so people are often regularly exposed.
Meanwhile, previous research has shown how rising temperatures, lower oxygen levels and heat stress, among other effects linked to climate change, may also worsen infertility.
Heat stress has been found to affect human hormones, and is linked to spermatogenesis in rodents and bulls.
Research shows temperature also plays a role in sex determination in fish, reptiles and amphibians.
The species has evolved to choose which sex it produces in part based on temperature, and the heating planet can “push it too far in one direction or the other, which overrides that evolutionary benefit”, Brander said.
Similarly, many endocrine disruptors may alter environmental sex determination.
The study set out some of the overlapping effects of chemical exposure and climate change across taxonomic groups, from invertebrates to humans.
In birds, for example, exposure to increased temperature, PFAS, organochlorines and pyrethroids may each individually cause abnormal sperm, increased fledgling mortality, abnormal testes and population decline.
“What happens if they’re exposed to more than one of those stressors at the same time? There has been little exploration of that question.
“Even if there have not been a lot of studies looking at these simultaneously, if you have two different factors that both cause the same adverse effect, then there’s a likelihood that they are going to be additive,” Brander said.
Katie Pelch, a senior scientist with the Natural Resources Defense Council nonprofit, who was not part of the study, said the authors had reviewed high-quality science.
She said she wanted to see more examples of the overlap in impacts, but agreed with the overall premise.
“It is likely [multiple stressors] would have an additive effect, at very least, even if they have different mechanisms of harm,” Pelch added.
The solution to the systemic problems would involve tackling climate change and reducing the use of toxic chemicals.
The study cites the global reduction in the use of DDT and PCBs achieved under the Stockholm Convention as an example of an effective measure, but Brander said much more is needed.
“There is enough evidence in both areas to act to reduce our impact on the planet,” she said.
Fertility
Researcher explores weight loss jab impact on PCOS

 Entrepreneur7 days ago
Entrepreneur7 days agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoThree sessions that show exactly where women’s health is heading in 2026

 Pregnancy4 weeks ago
Pregnancy4 weeks agoHow NIPT has evolved and what AI NIPT means in 2026

 News4 weeks ago
News4 weeks agoTwo weeks left to make your mark in women’s cardiovascular health

 Fertility2 weeks ago
Fertility2 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic

 Mental health1 week ago
Mental health1 week agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Menopause2 weeks ago
Menopause2 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 News4 weeks ago
News4 weeks agoCopper coil vs Mirena: Which is right for you?



























Pingback: Powerful short film from Future Fertility uniquely captures the emotional journey of fertility treatment