To receive the Femtech World newsletter, sign up here.
Entrepreneur
Could this start-up turn the tide on ovarian cancer?
Ovarian cancer causes more deaths than any other cancer of the female reproductive system

Nearly 225,000 women are diagnosed with ovarian cancer globally every year, but over 50 per cent of them will not survive. Could this biotech start-up have the solution?
March marks Ovarian Cancer Awareness Month – a chance to put ovarian cancer in the spotlight.
The disease causes more deaths than any other cancer of the female reproductive system and, according to the American Cancer Society, will kill in 2024 12,740 women in the US alone.
Despite this, awareness remains low. New data from Target Ovarian Cancer shows that, while progress has been made, things are not moving fast enough, with 40 per cent of UK women confusing cervical cancer and ovarian cancer.
“We don’t have enough awareness and advocacy around ovarian cancer to elevate the importance of the disease the way we do around breast cancer,” says Oriana Papin-Zoghbi, co-founder and CEO of the biotech start-up AOA Dx.
She believes this is directly linked to the low incidence of the disease – there are about 240,000 new cases of breast cancer in the US every year, compared to just under 20,000 of cases of ovarian cancer.
However, she says: “While we don’t see hundreds of thousands of cases, the percentage of women who die of ovarian cancer is significantly higher than almost any other gynaecological cancer.”
Ovarian cancer’s high fatality rate is largely attributed to a failure to recognise symptoms early on and a series of misconceptions around the disease, such as the idea that ovarian cancer is a “silent killer”.
Papin-Zoghbi agrees. Most of the things we hear about ovarian cancer, she says, are not actually true.
“Around 87 per cent of women with ovarian cancer experience symptoms early on, but they are misconstrued for different things.
“The vague abdominal symptoms that we hear about are commonly mistaken for conditions like endometriosis or irritable bowel syndrome because there is no diagnostic to differentiate them.”
Currently, the only way to confirm an ovarian cancer diagnosis is a tissue biopsy. Most women have a biopsy during an operation called laparotomy, where the surgeons make a large cut down the middle of the abdomen to take samples of tissue.
However, in some cases, doctors have to remove an ovary and have it tested for signs of cancer to confirm a diagnosis.
Papin-Zoghbi and her team at Boston-based AOA Dx are aiming to develop the first early-stage ovarian cancer liquid biopsy diagnostic test, which could significantly cut down fatalities and make the diagnosis process less invasive.
“The fact that 80 per cent of women are diagnosed with stage three and four ovarian cancer is unacceptable. Women need a much better chance,” she says.
“We are hoping that our solution will help elevate our understanding of ovarian cancer.”
The technology has shown over 90 per cent accuracy for detection of ovarian cancer and raises hopes for thousands of women globally. However, it will take time.
“There are certain that are not in our control,” says Papin-Zoghbi.
“It takes up every waking moment to build a company, but we have a strong team and we are very positive about the the path forward.
“We closed a US$17m financing round in October and we are currently focusing on building our lab, developing our product pipeline and conducting our clinical study to validate our technology. In the next couple of years, our aim is to focus on product development and clinical study data ahead of thinking about how we will bring the product to market.
“We’re optimistic – we know we can have a transformational impact on women’s health.”
News
Sun Pharma to acquire Organon in US$11bn deal
News
Women’s digital health market set to reach US$5.28 billion in 2026 – report
Entrepreneur
Future Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

Future Fertility Inc. has announced the closing of a US$4.1 million Series A financing round.
The round was led by M Ventures (the corporate venture capital arm of Merck KGaA, Darmstadt, Germany) and Whitecap Venture Partners, with participation from new investors Sandpiper Ventures, Gaingels, and Jolt VC.
The financing will accelerate Future Fertility’s commercial expansion into Asia-Pacific and support its entry into the United States, including planned FDA 510(k) clearance for additional products as part of a broader U.S. market entry strategy.
Proceeds will also advance the development of a broader AI platform, from egg assessment through to embryo transfer, designed to support clinicians, embryologists, and patients across the full IVF journey.
M Ventures and Whitecap have supported Future Fertility’s mission to translate AI innovation into meaningful clinical outcomes since the company’s earliest stages.
Oliver Hardick, investment director, M Ventures, said: “Future Fertility is addressing a critical unmet need in reproductive medicine with a differentiated AI platform grounded in clinical data and real-world workflow integration.
“We are excited to continue supporting the company and team because we believe its technology has the potential to improve decision-making for clinicians, bring greater clarity to patients, and help advance a more personalised standard of care in fertility treatment.”
Future Fertility’s AI platform addresses a long-standing gap in fertility care: historically, there has been no objective, clinically validated method for assessing egg quality (Gardner et al., 2025), despite it being one of the most important drivers of reproductive success.
The company’s suite of deep learning tools includes VIOLET™, MAGENTA™, and ROSE™, purpose-built for egg freezing, IVF, and egg donation respectively.
The tools are based on AI models trained and validated on more than 650,000 oocyte images and are deployed in over 300 clinics across 35 countries.
Rhiannon Davies, founding and managing partner, Sandpiper Ventures, said: “The best outcomes in fertility care globally come from better data and smarter tools. Future Fertility understands that, and they’ve built a platform that delivers on it.
“Sandpiper is proud to back a team turning rigorous science into real results for patients and clinicians alike.”
Partnerships with the world’s leading fertility networks – including IVI RMA and Eugin Group across Latin America and Europe, FertGroup Medicina Reproductiva in Brazil, and most recently announced Kato Ladies Clinic in Japan – reflect growing demand for objective, AI-powered oocyte assessment in fertility care. In the United States, ROSE™ is newly available under an FDA 513(g) determination.
Research shows that approximately 50 per cent of IVF patients do not understand their likelihood of success, and many discontinue treatment prematurely, even though cumulative success rates improve significantly with multiple cycles (McMahon et al., 2024).
By delivering earlier clarity on egg quality, Future Fertility’s tools support more informed conversations between clinicians and patients, helping set realistic expectations and guide decisions about next steps.
Future Fertility’s growing evidence base spans seven peer-reviewed publications in Human Reproduction, Reproductive BioMedicine Online, Fertility & Sterility, and Nature’s Scientific Reports, and more than 70 scientific abstracts accepted and presented with partner clinics at conferences worldwide.
Christine Prada, CEO, Future Fertility, said: “Fertility treatment is one of the most emotionally and physically demanding experiences a person can go through.
“Every patient deserves objective data, not just a best guess, to support better decisions at critical moments in their care.
“This funding means we can bring that clarity to more patients, in more countries, at a moment when it matters most.”
Find out more about Future Fertility at futurefertility.com

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
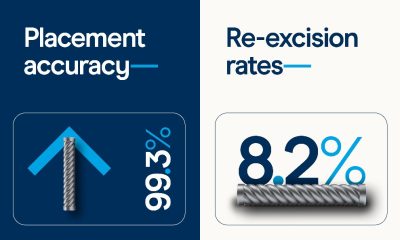
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Ageing3 weeks ago
Ageing3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 News4 weeks ago
News4 weeks agoResistance training has preventative effects in menopause, study finds

 Wellness2 weeks ago
Wellness2 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Insight2 weeks ago
Insight2 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now































