Cancer
Wrist cooling could help managing hot flash study finds

A cooling wristband device was found to reduce severe hot flash episodes by 46 per cent in breast cancer patients, prostate cancer patients and postmenopausal women, new research has shown.
The device reduced severe episodes by 41 per cent in breast cancer patients and 50 per cent in prostate cancer patients and postmenopausal women.
It also lowered daily hot flash frequency by 18 per cent.
Hot flashes – sudden, temporary sensations of body warmth, often with flushing and sweating during the day and night (night sweats) – are classed as vasomotor symptoms (VMS).]
These affect up to 80 per cent of perimenopausal and postmenopausal women, 80 per cent of ageing men receiving androgen deprivation therapy for prostate cancer, and between 50 and 80 per cent of breast cancer patients on hormone therapy.
The randomised, double-blind study at Boston University Chobanian & Avedisian School of Medicine involved 27 participants who experienced at least two moderate-to-severe hot flashes daily.
The group included 10 breast cancer patients, 12 prostate cancer patients and five postmenopausal women.
After two weeks of baseline recordings, participants used either the active cooling device or a non-cooling wristband for two weeks, then crossed over to the alternative device for an additional two weeks.
Hot flash frequency and severity were logged in diaries throughout.
Michael F. Holick, professor of medicine, pharmacology, physiology and biophysics at the school, said current treatments are limited.
Holick said: “Most non-hormonal pharmaceutical options for managing hot flashes are limited by significant side effects and hormone replacement therapy is not appropriate for breast and prostate cancer patients who were on, or who have been treated with, hormone deprivation therapy.
“This creates an urgent need for safe, effective, non-pharmaceutical interventions suitable for diverse patient populations experiencing debilitating VMS.”
VMS include hot flashes and night sweats, and are linked to sleep problems, cognitive difficulties, fatigue, increased pain and lower quality of life.
Holick said the wrist’s neurological sensitivity makes it an effective site for targeted cooling.
The device was developed using approaches applied in conditions such as motion sickness, Parkinson’s disease, Tourette’s syndrome and hand tremors.
He believes the cooling device signals the hypothalamus – the part of the brain that regulates body temperature – by triggering cooling neuron pathways.
This makes the body think it is cooling rapidly, shutting down the overheating signals that cause blood vessel dilation and sweating.
Holick noted that hot flashes affect more than comfort, influencing quality of life, decisions about cancer treatment and adding to wider economic and social costs.
Cancer
Common cholesterol drug shows ovarian cancer promise

A common cholesterol drug could help weaken a fluid shield that helps ovarian cancer tumours survive, early lab findings suggest.
The findings do not show the drug treats ovarian cancer. But they suggest changing the environment the cancer depends on could make it more vulnerable to existing treatment.
A federally funded study at Duke University School of Medicine found that ascites, a build-up of fluid in the abdomen, may do more than cause discomfort.
Doctors can drain ascites to ease pain, improve mobility and make breathing easier, but the fluid may also help cancer cells survive and spread. It occurs in 90 per cent of people with advanced ovarian cancer.
According to the study, ascites acts as a shield, helping cancer cells evade ferroptosis, a form of cell death.
Ferroptosis is a kind of cellular rusting. It happens when iron inside a cell reacts with certain fats, causing the cell membrane to break apart.
Many metastatic cancer cells, meaning cells that float freely through the abdomen looking for new places to grow, are naturally vulnerable to this kind of damage.
“Doctors have mostly viewed ascites as a symptom rather than an active driver of disease,” said Jen-Tsan Chi, professor in the department of molecular genetics and microbiology and co-leader of the Cancer Biology Program at the Duke Cancer Institute.
“We’ve learned it gives cancer a survival advantage, which fills a major gap in understanding how ovarian cancer spreads.”
Scientists bathed cancer cell lines and patient-derived tumour cells in ascites collected from patients and watched how they responded to ferroptosis triggers.
The fluid protected cancer cells by changing how they store fats and control iron levels, effectively blocking cell death.
The protection required only trace amounts, with as little as 2 per cent immersion shielding cancer cells from destruction.
“What surprised us was how selective this effect was,” said Yasaman Setayeshpour, first author and graduate student in molecular genetics and microbiology at Duke School of Medicine.
“Ascites didn’t protect the cancer cells from other well-known types of cell death, like apoptosis or necrosis, it only blocked ferroptosis.
“To figure out why, we broke ascites down into major parts, like lipids, proteins, and small molecules, and tested what happened when each was removed.
“When we took the lipids out, the protective effect disappeared. That told us lipids are the key reason ascites helps these cancer cells survive.”
But researchers found an unexpected helper in bezafibrate, an older cholesterol drug used to lower triglycerides by altering how the body processes fats.
The cholesterol drug restored sensitivity to ferroptosis, but only when ascites was present. On its own, the drug did not trigger cell death or slow tumour growth in mice.
The drug’s impact depended on the cancer’s surroundings, in this case the fat-rich fluid bathing the tumour. Researchers found that targeting this environment, using repurposed drugs like bezafibrate, could leave cancer cells more exposed to existing cancer treatments.
Chi said the finding could have implications beyond ovarian cancer. Other cancers, including colorectal and pancreatic cancers, can also spread within the abdominal cavity.
“This work shows how much the environment around a tumour matters,” Chi said.
“Biological fluids like ascites don’t just give cancer cells a place to move. They actively help drive how cancer spreads.”
Cancer
Artera receives FDA Clearance for breast cancer platform

Artera has won FDA clearance for ArteraAI Breast, its breast cancer platform for patients with early-stage HR-positive, HER2-negative invasive breast cancer.
ArteraAI Breast is the first and only FDA-cleared digital pathology-based risk stratification tool for breast cancer.
These FDA milestones come alongside recent CE marking for both the ArteraAI Prostate Biopsy Assay and the ArteraAI Breast Cancer Assay in the US and Europe.
“FDA clearance for ArteraAI Breast represents a significant expansion of our FDA-cleared AI platform in oncology,” said Andre Esteva, chief executive and co-founder of Artera.
“This milestone reflects the growing role of our technology across multiple cancer types. Breast cancer care is highly nuanced, with treatment decisions that depend on individualised risk.
“Our goal remains consistent across prostate and breast cancer, and beyond: to help clinicians translate complex data into more precise, personalised treatment decisions across the cancer journey.”
ArteraAI Breast generates an AI-derived risk score showing the likelihood of distant metastasis, meaning cancer spreading to another part of the body, in patients with early-stage HR-positive, HER2-negative breast cancer.
Using digitised histopathology images, which are scanned tissue sample images, alongside patient clinical variables, the model sorts patients into low-risk and high-risk groups based on a predefined risk score cut-off.
In early-stage HR-positive, HER2-negative breast cancer, deciding the right intensity of treatment can be complex because clinical and pathological factors vary. Artera said the tool is designed to support clinicians within established decision-making frameworks.
Data presented at the 2025 San Antonio Breast Cancer Symposium evaluated the model in early-stage breast cancer and demonstrated the potential to inform chemotherapy benefit in certain patient populations.
“This clearance represents an important advance on the road to personalising treatments for patients with early-stage breast cancer,” said Eric Winer, medical oncologist and director of the Yale Cancer Center.
“Using AI and digital pathology has the potential to streamline operational workflows, while creating a strong interdisciplinary linkage between oncology and pathology. This approach may further improve the clinicians’ ability to help patients make the best treatment decisions.”
ArteraAI Breast is designed to integrate directly into standard pathology workflows using routine surgical resection samples, without requiring additional tissue or separate specimen collection.
This approach allows the software to provide same-day results, enabling pathology laboratories to give clinicians patient-specific prognostic risk information alongside standard histopathology reports.
Diagnosis
Nurse celebrates role in trial that enhanced breast cancer surgery outcomes

A nurse who works with breast cancer patients has spoken of her pride after joining a trial that improved breast cancer surgery after her own diagnosis.
Heidi Jones, a 53-year-old staff nurse on the surgical day unit at Basildon Hospital, is a mother of two from Corringham in Essex and works with breast cancer patients daily.
She was diagnosed with breast cancer in 2025, the day after her birthday. She told ITV News Anglia that she had jumped at the opportunity to take part in the trial.
“I said yes because we need to get out there about the advancements in treatment and in surgery and medicines,” she said.
“Cancer is a big thing, so whatever you can do to improve the treatment of the cancer, I was all for it.”
Mid and South Essex NHS Foundation Trust was one of two UK centres taking part in the trial, alongside 21 others across the US, Canada and Austria.
The trial involved using a breast cancer locator, or BCL, a customised 3D mould matched to the unique dimensions of the patient’s tumour and breast.
The BCL is then placed over the patient during surgery, giving teams more detailed guidance on the tumour’s shape, size and location.
Results found a 34 per cent reduction in the number of second surgeries needed, and a 32 per cent reduction in cases where cancer remained after surgery.
Surgeon Wayne Chicken said the new technique could have a big impact on some patients.
“I’ve been working in breast surgery for 25 years, and breast surgery has changed radically in those 25 years,” he said.
“Now we’re trying to do less and less, the minimum necessary to control the disease, rather than big procedures and potentially over-treat cancers. This is doing the minimum necessary to treat the cancer.”
This actually uses the information from the MRI scans to plan, so the impact is less surgery and more likely to get it right in a single operation.
Using the BCL system, Jones’s breast cancer surgery was a success, with the tumour removed completely.
“I do talk about things quite openly, and I use it now when the ladies are coming in. I’ll tell them I’ve gone through it,” she said.
“It just gives them that little bit more to let them know someone else has come through [treatment] and has come through the other side.
“I feel proud that I actually took part in it. I feel privileged to have taken part in it and been asked to do it.
“I’m extremely glad it’s been really successful as well. I was lucky, I class myself lucky.”

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Diagnosis4 weeks ago
Diagnosis4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
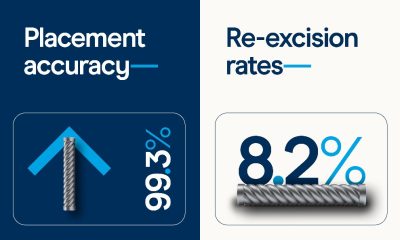
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both



























