Fertility
Round up: New insulin delivery technology supports healthier pregnancies and more

Femtech World explores the latest research and technology developments in the world of women’s health.
New insulin delivery technology supports healthier pregnancies
An international study has found new insulin delivery technology helps control glucose levels during pregnancy for those with Type 1 diabetes, which is crucial to the health of women and their newborns.
The technology, known as automated insulin delivery (AID), mimics a healthy pancreas. The system automatically adjusts the amount of insulin given by a pump in real-time, based on current and predicted glucose levels.
In a multicenter clinical trial, the researchers evaluated the impact of a hybrid closed-loop (HCL) insulin therapy treatment regime with standard insulin injections or an insulin pump that was not automated, along with continuous glucose monitoring.
“Keeping blood glucose in the optimal range for pregnancy is exceptionally challenging when someone has Type 1 diabetes, despite their best efforts and the support of dedicated health care clinics,” says Dr Denice Feig, MD, the study’s other co-principal investigator.
Risks associated with Type 1 diabetes in pregnancy can include increased chances of miscarriage, preeclampsia, which involves dangerous spike in blood pressure, and other significant health concerns.
Newborns of pregnant women with Type 1 diabetes are more likely to be born excessively large or early and have low blood glucose at birth and are at higher risk of birth defects.
“The study found this AID system worked in pregnancy.
“It resulted in a three hours per day improvement in the time spent in the desired glucose range compared to the standard delivery with insulin injections or regular insulin pumps,” says Donovan.
“This is very important because we have learned from other larger studies that every 72 minute per day increase, with glucose in the desired range during pregnancy, is associated with reduction in newborn complications.”
The AID system used in the study is known as a Tandem t:slim X2 insulin pump with Control-IQ technology.
The study found those using the AID spent more time in a healthy glucose level range and less time below and above the healthy range.
The improvement in blood sugar control was immediate and persisted throughout the pregnancy. These results were found at all 14 sites involved in the trial.
Widely-used technique for assessing IVF embryos may be flawed
A test deployed in many fertility clinics to assess the viability of embryos for use in IVF is likely to overestimate the number of embryos with abnormalities, a new study has suggested.
Using a new technique for imaging embryos in real time, a team led by scientists at the Loke Centre for Trophoblast Research, University of Cambridge, showed that abnormalities can arise at a later stage of embryo development than previously thought.
This means that the tests used in some clinics may be finding errors in cells that will go on to develop into the placenta – and abnormalities in placental cells are less likely to affect the health of the fetus.
When abnormalities are detected, the embryo may be deemed inviable and discarded, meaning patients may need to go through another cycle of treatment, which can prove costly.
So-called pre-implantation genetic testing for aneuploidy is a treatment ‘add on’ that may be offered to older women and those with a history of recurrent miscarriages or multiple IVF failures.
Researchers at the Loke Centre for Trophoblast Research, Cambridge, are interested in how early human embryos develop before implantation in the womb.
This is because in assisted conception, as many as nine in ten embryos fail to develop to a stage where they can be transferred to the womb.
To help understand development of the embryo at this early stage, Professor Niakan and colleagues, in collaboration with researchers at the Francis Crick Institute, developed a new, state-of-the-art method for watching embryos live in high resolution.
The new imaging technique involves tagging DNA inside the cell nucleus with a fluorescent protein, making it visible under a microscope.
The researchers then use an imaging technique known as light-sheet microscopy to observe the embryos in 3D as they developed without damaging them.
Of the 13 embryos analysed by the team, 10 per cent of the cells contained chromosomal abnormalities.
These arose from problems when DNA was being copied between cells, for example when chromosomes did not move properly during division or when a cell divides into three, rather than two.
Because these abnormalities arise at a relatively late stage of the embryo’s development, they appear in the outer layer of the blastocyst, which develops into the placenta – and it is from this layer that biopsies are taken for pre-genetic testing for aneuploidy.
Professor Niakan’s team is now studying cells in the inner layer to see whether such spontaneous abnormalities can also arise there.
In mice, fertility treatments linked to higher mutations than natural conception
Mice pups conceived with IVF in the lab have slightly increased rates of DNA errors, or mutations, compared to pups conceived naturally.
While the results from this new study do not directly apply to humans, they highlight the importance of understanding how fertility treatments affect an offspring’s DNA.
For the study, the researchers compared genome sequences of lab mice conceived naturally and mice conceived through assisted reproductive technologies, including hormone treatments, IVF, and embryo transfer.
They discovered pups born through these fertility treatments had about 30 per cent more new single-nucleotide variants, or tiny changes in DNA sequences.
Nucleotides are DNA’s building blocks or “letters.”
Arranged in specific sequences, these letters compose the instructions cells use to grow and function.
Single-nucleotide variants are simply genetic differences (or mutations) involving a change in just one DNA letter. They can occur when cells replicate their DNA.
The mutations observed in the study are unlikely to be harmful.
Scientists estimate that fewer than 2 per cent of new mutations arising in a genome are deleterious or have an impact on an individual’s phenotype or disease susceptibility.
The mutations appeared spread across the genome, rather than clustered in particular genes.
The timing of when these new mutations appeared in early embryos also looked similar between fertility-treated and natural groups, implying that fertility treatment increases the overall chance of new DNA changes but does not impact when they occur during development.
Even with a 30 per cent increase in new mutations, the absolute number of harmful new mutations per mouse remains low.
For about every 50 mice conceived with IVF, scientists expect roughly one additional harmful DNA change compared to natural conception.
That is one problematic change out of many possible ones, since the mouse genome is about 2.7 billion DNA letters long.
A similar effect is expected if the male parent’s age increased by about 30 weeks, the researchers explained, since paternal age is a major driver of mutation rates in mammals.
The biological mechanisms underlying these genetic changes are not clear.
Further research is needed to study whether the new mutations come from a specific step in the IVF process or from the combined effects of several steps.
One possible factor is the use of hormone treatments that stimulate the ovaries, since these hormones push eggs to restart meiosis, a stage of cell division known to be prone to mistakes.
Other aspects of the fertility treatment protocol could also play a role, such as physical handling of embryos or the chemical conditions of the lab culture environment.
The study does not show whether the same effect happens in humans. Fertility procedures vary between mice and humans, and both have different reproductive biology.
For example, mice do not menstruate. Also, people seeking IVF will likely encounter environmental factors that may already have affected their genetics.
Fertility
Genetic carrier screening before pregnancy: What to know

Article produced in association with London Pregnancy Clinic and Jeen Health
For the majority of couples planning a pregnancy, genetic testing is not something they think about until a problem arises.
Pre-conception genetic carrier screening challenges this approach by identifying risk before pregnancy begins.
As panel sizes have grown and at-home testing options have become widely available, carrier screening is transitioning from a niche clinical referral into a mainstream component of reproductive planning.
What Carrier Screening Tests For
Being a carrier of a genetic condition means carrying one copy of a variant in a gene associated with that condition, without being affected by it.
In most cases, carriers are entirely unaware of their status.
The clinical significance of carrier status emerges when both members of a couple carry a variant in the same gene: in this scenario, each pregnancy carries a one in four chance of resulting in a child who inherits two copies of the variant and is affected by the condition.
The conditions most frequently included in expanded carrier screening panels include cystic fibrosis, spinal muscular atrophy (SMA), fragile X syndrome, sickle cell disease, and a range of metabolic and enzyme deficiency disorders.
The Beacon 787 carrier test, offered by Jeen Health, screens for 787 conditions from a single sample, making it one of the most comprehensive panels currently available to UK families.
Who Is Most Likely to Benefit
Any couple planning a pregnancy can consider carrier screening. It is particularly relevant for:
- Couples with a family history of a known inherited condition
- Those from populations with higher carrier frequencies for specific conditions, including Ashkenazi Jewish, South Asian and African communities
- Couples pursuing fertility treatment, where genetic information informs treatment planning
- Those who wish to have the most complete picture of their reproductive health before conception
Importantly, being a carrier of a condition does not mean a child will be affected. It means there is a defined statistical risk that can be quantified, discussed and planned for with appropriate clinical support.
How the Test Is Performed
Carrier screening is typically carried out on a blood or saliva sample.
For at-home options such as the testing offered by Jeen Health, a cheek swab collection kit is dispatched to the patient, the sample is returned by post, and results are delivered digitally within a defined turnaround period.
In-clinic carrier testing may use a blood draw and provides the advantage of immediate access to a clinical consultation at the point of result delivery.
London Pregnancy Clinic offers genetics counselling through its partnership with Jeen Health, allowing couples to receive and contextualise carrier test results with expert support.
Genetic counselling before and after testing is recommended by Genomics England as a standard component of any genomic testing pathway.
What Happens If Both Partners Are Carriers
If both partners are identified as carriers for the same autosomal recessive condition, they are typically offered further counselling to discuss their options.
These may include proceeding naturally with an awareness of the risk, using prenatal diagnosis (CVS or amniocentesis) during pregnancy to test the fetus, or pursuing preimplantation genetic testing (PGT) in the context of IVF, which allows unaffected embryos to be selected before transfer.
The purpose of identifying carrier status before pregnancy is to give couples time to consider these options without the added pressure of an ongoing pregnancy.
Knowledge of carrier status does not remove reproductive choices; it expands the information available when making them.
The Role of Pre-Conception Services
Carrier screening sits within a broader category of pre-conception care that includes fertility assessments, general health optimisation and, where relevant, management of existing conditions before pregnancy begins.
London Pregnancy Clinic offers pre-conception services encompassing fertility investigations, genetics counselling and carrier testing as part of an integrated 0th trimester approach, allowing couples to address genetic and clinical risk factors before their pregnancy starts rather than after.
Disclaimer: This article is produced for informational purposes only and does not constitute medical advice, diagnosis or treatment.
Clinical guidance referenced reflects published NHS, NICE and RCOG standards as at March 2026. Individual circumstances vary; readers are advised to consult a qualified healthcare professional before acting on any information in this article.
This piece was produced in association with London Pregnancy Clinic and Jeen Health, which provided background clinical information for editorial purposes.
Hyperlinks to external sources are included for reference only and do not represent an endorsement of any product, service or organisation.
Fertility
Fertility clinic named London finalist in UK StartUp Awards

A London-based fertility clinic has been shortlisted for a startup award.
Plan Your Baby was shortlisted as a London finalist for Innovative Startup of the Year at the UK StartUp Awards.
Plan Your Baby is a new generation fertility and pregnancy telehealth clinic that provides fertility treatment and and-to-end pregnancy clinical monitoring and psychological support.
The company said on LinkedIn: “Being recognised in a city as competitive as London is meaningful for our team.
“The award is judged by industry experts and reflects the growing need for fertility care that is structured, transparent, and centred around the patient.
“Many people come to us looking for clarity in what can often feel like a complex process.
“Our focus has been to make each step easier to understand and easier to access.”
Plan Your Baby founder Marija Skujina was inspired to launch the company after working at the highest level in private fertility clinics and realising the impact that the traditional approach to fertility treatment was having on clients.
She told Femtech World in a 2023 interview: ““Fertility support is not just a medical procedure, it’s physical, mental, and emotional too.
“That’s why I launched Plan Your Baby: to help parents conceive in a fully supported and holistic manner.”
The UK StartUp Awards aim to ‘recognise the achievements of amazing individuals who have had a great idea, spotted the opportunity and taken the risks to launch a new product or service.’
If selected as the regional winner, Plan Your Baby will go on to the national final at Ideas Fest this September.
Previous winners include Magic AI, makers of a wall-mounted AI fitness mirror that acts as a personal trainer, and EnsiliTech, a medtech startup developing innovative health technology solutions at the intersection of engineering and healthcare.
Fertility
Future Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic

Future Fertility, a Toronto-based health technology company specialising in AI-powered fertility insights, has entered the Japanese market through a new commercial partnership with Kato Ladies Clinic — a globally recognised leader in IVF research and advancing clinical fertility care.
The collaboration marks Future Fertility’s first partnership in Japan and reflects growing global demand for technologies that bring greater objectivity and personalisation to fertility care.
Kato Ladies Clinic will integrate the company’s AI-powered oocyte (egg) quality assessment tools into its clinical workflows, with the aim of supporting more informed treatment planning and patient counselling across IVF and egg freezing cycles.
“At Kato Ladies Clinic, we are committed to advancing fertility care through innovation while maintaining a strong focus on individualised, patient-centred treatment,” said Keiichi Kato, chief executive officer.
“Partnering with Future Fertility enables us to integrate objective, data-driven insights into our clinical approach and better support our patients in making informed decisions.”
Future Fertility’s platform analyses images of oocytes using artificial intelligence trained and validated on a dataset of more than 650,000 unique oocyte images.
The technology is already in use at more than 300 clinics across more than 35 countries, helping clinicians better understand the developmental potential of individual eggs and provide patients with more personalised insight earlier in their treatment journey.
From Research Collaboration to Clinical Adoption
The partnership between Future Fertility and Kato Ladies Clinic began as a scientific research collaboration in 2024, marking the first use of AI-powered oocyte quality assessment in Japan.
The collaboration not only validated the technology in a new patient population and across diverse clinical protocols — including minimal stimulation cycles —but also resulted in a peer-reviewed publication in Reproductive BioMedicine Online (RBMO) and a poster abstract presentation at ESHRE 2025.
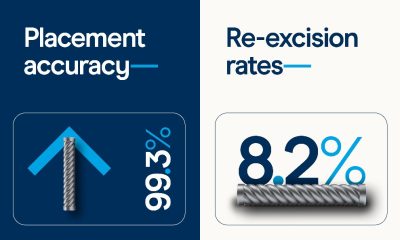
The joint research explored how AI-derived oocyte quality scores relate to early embryonic development and overall treatment outcomes. In a retrospective study conducted at Kato Ladies Clinic, researchers analysed nearly 2,800 mature oocytes across more than 1,300 ICSI cycles, linking image-based assessments of egg quality to key developmental milestones.
The study demonstrated that lower AI scores were associated with reduced fertilization rates, delays, and abnormalities in early embryo development, increased developmental errors, and lower-quality blastocyst formation.
Notably, the researchers also found that cumulative oocyte scores were a stronger predictor of live birth outcomes than the number of eggs retrieved — underscoring the importance of assessing egg quality alongside quantity.
“Our collaboration with Future Fertility has demonstrated how artificial intelligence can uncover meaningful biological differences between oocytes that were previously difficult to quantify,” said Kenji Ezoe, senior scientist.
“Bringing this technology into routine clinical use is an important step toward translating research into improved patient outcomes.”
Future Fertility’s VP of clinical embryology & scientific operations, Jullin Fjeldstad, noted that the findings provide important clinical validation.
“Our joint research with Kato Ladies Clinic has shown how AI-based oocyte assessment can be directly linked to numerous embryo development outcomes, from fertilization through early developmental milestones and blastocyst formation,” she said.
“We are excited to see this work translated into clinical practice.”
Growing Demand for Fertility Care in Japan
The partnership comes at a time when demand for fertility treatment in Japan continues to rise.
The country performs over 450,000 fertility treatment cycles annually, making it one of the largest markets globally. Delayed childbearing and evolving societal trends have also contributed to increasing interest in egg freezing.
As patients seek more clarity and personalization in their care, tools that provide earlier insight into reproductive potential are gaining traction.
“Entering the Japanese market with a partner like Kato Ladies Clinic is a significant step forward for our global commercial strategy,” said Rafael Gonzalez, Future Fertility’s VP of global sales & strategy.
“It reflects the growing demand for technologies that support more transparent, data-driven fertility care across diverse healthcare systems.”
Expanding a Global Footprint
Founded in 1993, Kato Ladies Clinic is known for its pioneering work in natural and minimal stimulation IVF and has long been a leader in clinical innovation in Japan.
For Future Fertility, the partnership represents both a geographic expansion and a continuation of its broader mission to bring AI-driven insights into routine fertility care.
“We are proud to partner with Kato Ladies Clinic, a globally respected leader in IVF and a pioneer in reproductive medicine in Japan,” said Future Fertility’s CEO, Christy Prada.
“This partnership represents an important milestone as we expand into Asia and continue our mission to bring objective, personalised insights into fertility care worldwide.”
Future Fertility develops AI-powered tools designed to generate personalised insights across the fertility journey.
Its flagship oocyte assessment technologies analyse egg images to provide objective, individualised measures of egg quality, supporting treatment planning, patient counselling, and clinical decision-making in egg freezing and IVF, while also enabling more data-driven approaches to donor egg distribution and quality assurance.
As fertility care continues to evolve, collaborations like this one are helping shape a new standard — one that emphasises earlier insight, greater transparency, and more personalised decision-making for patients navigating increasingly complex reproductive journeys.

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoThree sessions that show exactly where women’s health is heading in 2026

 Pregnancy3 weeks ago
Pregnancy3 weeks agoHow NIPT has evolved and what AI NIPT means in 2026

 Menopause4 weeks ago
Menopause4 weeks agoWatchdog bans five ads for women’s heath claims

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoWHIS USA 2026 announces first ticket release for landmark Women’s Health Innovation Summit

 Menopause4 weeks ago
Menopause4 weeks agoMenopause has no lasting impact on cognition, research finds

 News3 weeks ago
News3 weeks agoTwo weeks left to make your mark in women’s cardiovascular health

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoQ1 momentum: Female founders are advancing, but the system still hasn’t caught up

 News4 weeks ago
News4 weeks agoEndometriosis firm wins NIH prize

























10 Comments