Diagnosis
Breast cancer screening catches more than 60,000 cases, but uptake declining

Screenings for breast cancer in the UK have caught more than 60,000 cases, however, participation in cancer screening is struggling to return to pre-Covid levels.
In England, breast cancer screenings are conducted to help prevent cancer or catch cancer at early stage.
The new insights from Cancer Research UK reveal that from 2019 to 2023, these screenings have diagnosed around 62,600 cases of cancer.
Despite this, the insights show that uptake for breast cancer screening is struggling to return to pre-Covid levels, with levels down from 71.1 per cent in 2018 to 2019 to 64.6 per cent from 2022 to 2023.
Cancer Research UK is now calling on the UK Government to work with the NHS to make it easier for people who are eligible to access screening.
Michelle Mitchell, chief executive of Cancer Research UK, said: “Every year, screening programmes help spot thousands of cancer cases across England. This is testament to the research that made these programmes possible, and the hardworking NHS staff who deliver them.
“But there’s so much more that can be done. Making screening more digital, and improving data collection to know who is and isn’t coming forward, will help to target people who currently miss their appointments, but would like to attend. This is essential – better access to screening means better chances of catching cancer early.”
Reasons some people are not attending screenings include not having symptoms, being too embarrassed to attend, and the experience or anticipation of pain for breast screening.
The insights also reveal that people from ethnic minority groups and people from lower income backgrounds are less likely to attend screening.
Hormonal health
Tampons could track MS nerve damage, study suggests

Menstrual fluid collected from tampons could one day provide a simple, non-invasive way to measure a biomarker of nerve damage and potentially track disease activity in neurological conditions such as multiple sclerosis (MS), new research suggests.
Because neurofilament light chain, or NfL, has emerged as a promising biomarker of MS, detecting it in menstrual fluid raises the possibility of monitoring disease activity through the natural monthly cycle of menstruation.
Researchers at Nextgen Jane, in collaboration with Siemens Healthineers, found that NfL, a protein released when nerve cells are damaged, can be reliably detected in tampon-collected menstrual samples.
“Finding that NfL tracks with estrogen levels in menstrual fluid, independent of how much blood is in the sample, tells us there is real biology here, not just contamination,” said Ridhi Tariyal, chief executive and co-founder of Nextgen Jane.
“That changes what this specimen means for neurology.”
In MS, the immune system mistakenly attacks healthy parts of the brain and spinal cord, causing inflammation and damage that can lead to symptoms such as fatigue, numbness, muscle weakness, and problems with balance or vision.
Confirming a diagnosis of MS usually requires a combination of physical and neurological examinations, MRI scans to check for brain and spinal cord damage, and lab tests.
These can include detecting certain proteins in cerebrospinal fluid, the fluid that surrounds the brain and spinal cord, which may indicate inflammation in the brain or spinal cord.
After diagnosis, patients are usually monitored through clinical assessments and routine MRI scans, which help doctors detect changes in disease activity and determine whether treatments are working.
However, MRI assessments can be costly and are usually done once or twice a year, which can prevent doctors from spotting early changes and making timely treatment adjustments.
Because of these challenges, researchers have long sought cost-effective, more accessible biomarkers that could help detect MS earlier, monitor disease activity over time, and evaluate treatment response.
One of the most promising candidates is NfL, a protein found in nerve cell fibres that is released into the bloodstream and cerebrospinal fluid when nerve cells are injured.
To explore whether menstrual fluid could serve as a source for detecting this biomarker and, more broadly, as a non-invasive specimen for monitoring neurological, hormonal and inflammatory signals, researchers analysed 99 tampon-collected menstrual fluid samples from 91 participants.
They used Siemens Healthineers’ highly sensitive NfL assay on its automated testing platform. The team also measured hormonal and inflammatory molecules.
NfL was detected in 98 of the 99 menstrual fluid samples analysed, suggesting the biomarker can be reliably measured in tampon-collected samples.
The researchers also found that NfL levels were associated with estradiol levels, a form of the hormone oestrogen, and that this relationship remained significant even after adjusting for differences in blood content between samples.
By comparison, levels of inflammatory markers were more strongly linked to blood content itself.
According to the researchers, this suggests NfL detection was not merely the result of blood contamination, but may reflect biologically meaningful changes that could potentially be tracked over time through routine menstrual sampling.
Building on these findings, Nextgen Jane is now planning prospective studies to investigate whether menstrual NfL and other neurological proteins can be used to track disease activity over time in conditions such as MS.
“The menstrual cycle provides a built-in longitudinal framework: the same individual, the same biological process, month after month,” said Stephen Gire, chief scientific officer at Nextgen Jane.
“Coupling the NextGen Jane platform with Siemens Healthineers’ highly sensitive NfL assay gives us a path to study neurological biomarker trajectories in a way that has not been possibe before.”
Diagnosis
Artera receives FDA Clearance for breast cancer platform

Artera has won FDA clearance for ArteraAI Breast, its breast cancer platform for patients with early-stage HR-positive, HER2-negative invasive breast cancer.
ArteraAI Breast is the first and only FDA-cleared digital pathology-based risk stratification tool for breast cancer.
These FDA milestones come alongside recent CE marking for both the ArteraAI Prostate Biopsy Assay and the ArteraAI Breast Cancer Assay in the US and Europe.
“FDA clearance for ArteraAI Breast represents a significant expansion of our FDA-cleared AI platform in oncology,” said Andre Esteva, chief executive and co-founder of Artera.
“This milestone reflects the growing role of our technology across multiple cancer types. Breast cancer care is highly nuanced, with treatment decisions that depend on individualised risk.
“Our goal remains consistent across prostate and breast cancer, and beyond: to help clinicians translate complex data into more precise, personalised treatment decisions across the cancer journey.”
ArteraAI Breast generates an AI-derived risk score showing the likelihood of distant metastasis, meaning cancer spreading to another part of the body, in patients with early-stage HR-positive, HER2-negative breast cancer.
Using digitised histopathology images, which are scanned tissue sample images, alongside patient clinical variables, the model sorts patients into low-risk and high-risk groups based on a predefined risk score cut-off.
In early-stage HR-positive, HER2-negative breast cancer, deciding the right intensity of treatment can be complex because clinical and pathological factors vary. Artera said the tool is designed to support clinicians within established decision-making frameworks.
Data presented at the 2025 San Antonio Breast Cancer Symposium evaluated the model in early-stage breast cancer and demonstrated the potential to inform chemotherapy benefit in certain patient populations.
“This clearance represents an important advance on the road to personalising treatments for patients with early-stage breast cancer,” said Eric Winer, medical oncologist and director of the Yale Cancer Center.
“Using AI and digital pathology has the potential to streamline operational workflows, while creating a strong interdisciplinary linkage between oncology and pathology. This approach may further improve the clinicians’ ability to help patients make the best treatment decisions.”
ArteraAI Breast is designed to integrate directly into standard pathology workflows using routine surgical resection samples, without requiring additional tissue or separate specimen collection.
This approach allows the software to provide same-day results, enabling pathology laboratories to give clinicians patient-specific prognostic risk information alongside standard histopathology reports.
Diagnosis
Nurse celebrates role in trial that enhanced breast cancer surgery outcomes

A nurse who works with breast cancer patients has spoken of her pride after joining a trial that improved breast cancer surgery after her own diagnosis.
Heidi Jones, a 53-year-old staff nurse on the surgical day unit at Basildon Hospital, is a mother of two from Corringham in Essex and works with breast cancer patients daily.
She was diagnosed with breast cancer in 2025, the day after her birthday. She told ITV News Anglia that she had jumped at the opportunity to take part in the trial.
“I said yes because we need to get out there about the advancements in treatment and in surgery and medicines,” she said.
“Cancer is a big thing, so whatever you can do to improve the treatment of the cancer, I was all for it.”
Mid and South Essex NHS Foundation Trust was one of two UK centres taking part in the trial, alongside 21 others across the US, Canada and Austria.
The trial involved using a breast cancer locator, or BCL, a customised 3D mould matched to the unique dimensions of the patient’s tumour and breast.
The BCL is then placed over the patient during surgery, giving teams more detailed guidance on the tumour’s shape, size and location.
Results found a 34 per cent reduction in the number of second surgeries needed, and a 32 per cent reduction in cases where cancer remained after surgery.
Surgeon Wayne Chicken said the new technique could have a big impact on some patients.
“I’ve been working in breast surgery for 25 years, and breast surgery has changed radically in those 25 years,” he said.
“Now we’re trying to do less and less, the minimum necessary to control the disease, rather than big procedures and potentially over-treat cancers. This is doing the minimum necessary to treat the cancer.”
This actually uses the information from the MRI scans to plan, so the impact is less surgery and more likely to get it right in a single operation.
Using the BCL system, Jones’s breast cancer surgery was a success, with the tumour removed completely.
“I do talk about things quite openly, and I use it now when the ladies are coming in. I’ll tell them I’ve gone through it,” she said.
“It just gives them that little bit more to let them know someone else has come through [treatment] and has come through the other side.
“I feel proud that I actually took part in it. I feel privileged to have taken part in it and been asked to do it.
“I’m extremely glad it’s been really successful as well. I was lucky, I class myself lucky.”

 Fertility3 weeks ago
Fertility3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
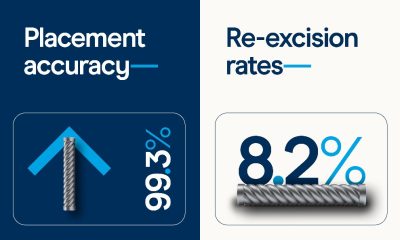
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoFlora Fertility closes US$5m seed round

 Menopause3 weeks ago
Menopause3 weeks agoResistance training has preventative effects in menopause, study finds




























1 Comment