Insight
The future of women’s health is in collaboration
By Jessica Aird, manufacturing manager at Abingdon Health

Abingdon’s Health’s manufacturing manager, Jessica Aird, takes a look at all things women’s health with a particular focus on how the organisation in which she operates so effectively – an expert lateral flow CRO & CDMO business – works together to deliver innovation and change in this area on a daily basis.
On March 8, 2021, the government launched a call for evidence to inform the development of England’s first Women’s Health Strategy, with two main aims:
- First, to improve the way in which the health and care system listens to women, and to reset our approach to women’s health by placing women’s voices at the centre of this work.
- Second, to improve women’s health outcomes.
As part of this launch, the government called for organisations with expertise in women’s health to submit written evidence into the following:
- Current medicines and medical devices.
- Geographical differences in women’s life expectancy and access to services.
Women’s specific health can be categorised in two main stems of health needs: general and reproductive specific. The diagram below explores these two stems and how these needs change during the course of a woman’s life.

Two main stems of women’s health needs (Department of Health and Social Care, 2021)
Abingdon Health’s vision is to become the leading rapid test business globally and share our mission – to improve life by making rapid testing accessible to all – with all industries. This includes improvement to current women’s health testing solutions through the development and manufacture of new, innovative, and creative medical devices.
The role of Abingdon Health in the improvement of women’s health strategy does not just lie within the manufacturing process.
There are many supporting departments working in unison to ensure any devices which successfully enter the market are the best solution available for women to further improve their knowledge and understanding of their health and wellbeing.
R&D
Abingdon’s R&D team are either approached by commercial on behalf of a customer with a bespoke women’s health testing solution, or the idea comes from within the organisation.
The process of progressing the idea from a concept to small scale manufacture relies heavily on R&D. The team must develop the device with key focuses on:
- Selecting the most efficient and appreciate materials
- Testing for cross-reactivity
- Optimisation and repeatability of performance
- Scalability considerations are also considered at this stage.
Technical transfer
The key role of the technical transfer department is to take each R&D-proven small-scale manufacturable assay to a full-scale production batch; this could be up to 30,000 devices per batch run. This is achieved through the following processes:
- Understanding potential failure modes, risk to assay performance and what current controls are in place in order to mitigate the risks. This is done predominantly by the technical transfer team, but also reviewed and adjusted collaboratively through input of many departments, including production, quality control, quality assurance and technical specialists.
- Transferring the processes from small scale equipment to large production equipment within the production laboratories whilst understanding the how the process parameters must change to keep product performance optimised and results, sensitivity and specificity within range at scale.
- Robustness testing of assay capacity and the assay performance in terms of scaling up the batches. This is done in order to understand the limitations of an assay. Robustness testing typically includes studies of treated materials, stability on part-processed components and stacked tolerances to determine optimal performance going forward.
Regulatory
Product regulatory compliance plays a significant role in bringing the concept of a new product in women’s health to reality and onto the market as fully reproducible manufactured product.
The regulatory team are part of development of the new assay from the very beginning; this is crucial as it helps the team to understand the regulatory requirements and anticipate where potential Quality Assurance and Regulatory Affairs (QARA) issues may arise.
The implications of a non-conformance at any stage of product development can cause significant timing delays, product redesign requirements and increased costs, so it is essential for QARA functions to have input at all stages of development of a new product.
Regulatory approvals are the last step before product launch to market. Obtaining official approval for a product to go to market, particularly in the case of a medical device, requires immense planning with manufacturing, quality assurance and commercial to ensure the product to be launched is viable and has the best chance of success in its newly obtained distribution regions and channels.
Customer services and commercial
Keeping the customer intimately involved in the product development process is essential to successful project and product performance and launch onto market.
The commercial team and customer services functions work hand in hand with both the new product development internal project team members and customer stakeholders to ensure direct interaction between both parties is maintained and expectations are managed.
Ensuring customers sign off at project stage gates, review quality control data and approve operating procedures and product process parameters all require commercial and customer service organisation.
Production and shipping
Once the product is ready for launch, customer service work closely with our quality, regulatory and warehousing teams to ship products to customer on time and in full. This can involve booking shipments with couriers, completing the product release process, and managing customer expectations of timelines.
In summary, the future of women’s health is indeed in collaboration: working closely cross-departmentally to take a concept or idea through to developing a product ready for marketing which can improve women’s access to healthcare, all the way through the research and development to technical transfer and production, is critical to success.
Working collaboratively will ensure better access to healthcare for women and inspire others to come forward with innovative solutions to everyday issues in women’s healthcare today and throughout the course of our lives.
To find out more, visit abingdonhealth.com.

Insight
Common cancer marker may play active role in preventing the disease, study finds
Ki-67, a protein used to measure tumour growth, may also help prevent chromosome errors that drive cancer, a study suggests.
The findings could change how scientists view Ki-67, a marker commonly used in breast cancer and other tumours to assess how quickly cancer cells are growing.
Researchers found the protein may help preserve genome stability by maintaining the structural integrity of centromeres, key parts of chromosomes that help ensure DNA is shared correctly during cell division.
The research was led by professor Paola Vagnarelli at Brunel University of London in collaboration with scientists at the University of Edinburgh and the Technical University of Berlin.
Professor Vagnarelli said: “Doctors already measure Ki-67 to see how aggressive a cancer might be. But our results suggest it is actually helping maintain genome stability.
“That means it may be more than a marker. It could potentially also be a therapeutic target.”
The study examined three proteins that attach to chromosomes during cell division and help rebuild the molecular system that tells each new cell what kind of cell it is.
Every human cell carries identical DNA. What makes a liver cell different from a brain cell is which genes are switched on and which are kept inactive.
When a cell divides, that entire system of switches must be rebuilt. The three proteins involved in this process were Ki-67, Repo-Man and PNUTS.
Vagnarelli’s team developed a method that individually removes each protein from a living cell at the precise point of division. Older techniques could not isolate that moment cleanly.
They found that cells rely on all three proteins to reset themselves after division, but each failed in a different way when removed.
Without PNUTS, gene activity spiralled out of control and thousands of genes switched on at once.
Without Repo-Man, cells escaped safety checkpoints that usually stop damaged or abnormal cells from continuing to divide.
“What we didn’t expect was how clean the separation was,” said Vagnarelli.
Each protein fails in its own specific way. There is no redundancy, no safety net. Which means there are three separate points at which this process can go wrong.
“When the system breaks down, cells can emerge with the wrong number of chromosomes. That condition, called aneuploidy, is seen in disorders such as Down syndrome and in many cancers.
“We also found that these chromosome errors can trigger inflammatory signals inside the cell.”
Aneuploidy means a cell has too many or too few chromosomes, which can disrupt normal growth and function.
Inflammatory signals are chemical messages that can make a cell behave as if it is responding to injury or infection.
“These cells behave almost as if they are under attack,” said Vagnarelli.
“The immune response switches on because the genome is unstable.
“That link between chromosome imbalance and inflammation could help explain patterns we see in several diseases.”
The researchers said the findings may help cancer scientists better understand how chromosome instability, loss of gene regulation and cells dividing before they are ready contribute to tumour growth.
They said understanding the normal machinery that prevents these errors may help researchers find ways to push cancer cells into making mistakes they cannot survive.
“We now have a clearer map of the machinery that resets the cell after division,” said Vagnarelli.
“That knowledge gives us a starting point for thinking about new therapeutic approaches.”
Hormonal health
PCOS renamed after decade-long campaign to end ‘cyst’ misconception
News
The RESIL-Card tool launches across Europe to strengthen cardiovascular care preparedness against crises

By Women As One
Women As One is proud to have contributed to the development of the RESIL-Card tool as an active Advisory Board member, ensuring that gender equity and the perspectives of women cardiologists were embedded from the outset.
Through strategic input on the project’s design, formal support of its EU4Health funding application, and ongoing participation in advisory activities, Women As One has helped shape both the direction and implementation of this initiative.
By amplifying awareness, facilitating engagement from our global community, and advocating for inclusive representation, we have worked to ensure that RESIL-Card reflects the diverse realities of cardiovascular care and supports more equitable, resilient health systems in times of crisis. Read more about our involvement here.
On the European Day for Prevention of Cardiovascular Risk (March 14), the RESIL-Card consortium proudly announces the official launch of the RESIL-Card tool, a free online resource designed to help hospital cardiovascular professionals and other stakeholders assess and strengthen the resilience of their care pathways — ensuring that lifesaving care remains accessible even during times of crisis.
Available now at https://www.wecareabouthearts.org/resil-card/online-tool/, the RESIL-Card tool offers a structured self-assessment framework for evaluating the preparedness of cardiovascular services and identifying concrete actions to maintain continuity of care when health systems face disruption.
“Cardiovascular care must remain uninterrupted regardless of the challenges health systems face,” said Professor William Wijns, Research Professor in Interventional Cardiology, University of Galway, Ireland, and We CARE – RESIL-Card Coordinator.
“The RESIL-Card tool provides healthcare teams with a practical way to assess preparedness, identify improvement opportunities, and ultimately ensure that patients continue to receive lifesaving care when it matters most.”
Why the RESIL-Card tool was developed
Cardiovascular diseases remain the leading cause of death in Europe, making the continuity and resilience of care pathways a public health priority.
Despite advances in diagnosis and treatment, recent crises – from pandemics to geopolitical instability – have exposed the vulnerability of healthcare systems.
In today’s increasingly uncertain health landscape and global environment, proactive preparedness is no longer optional – it is essential.
The RESIL-Card tool was developed as part of an EU4Health-funded initiative to support organisations providing lifesaving cardiovascular care in strengthening their preparedness, improving coordination, and safeguarding patient outcomes in times of disruption.
The initiative focuses on practical resilience strategies to help health systems anticipate challenges rather than simply react to them.
“Healthcare systems today operate in an increasingly complex and unpredictable environment,” said Ariadna Sanz, Health Policy Manager at the Catalan Health Service (CatSalut).
“Tools like RESIL-Card help shift the focus from responding to crises toward proactively building strong, adaptable cardiovascular care pathways that protect patients over the long term.”
A collaborative and evidence-based methodology
The RESIL-Card tool is grounded in a robust, multidisciplinary development process involving cardiovascular experts, healthcare professionals, public health specialists, patient organisations, and policy stakeholders from across Europe.
Its development combined comprehensive literature reviews and analysis of existing preparedness frameworks with extensive stakeholder consultations and co-creation workshops. Real-world insights from healthcare providers and patient representatives were integrated throughout the process to ensure the tool reflects the practical realities of cardiovascular care delivery. The methodology also included iterative testing and validation phases, allowing the consortium to refine the tool and ensure it is both scientifically rigorous and practical for everyday use.
“From the outset, RESIL-Card was co-created with clinicians, patient representatives, and health system experts to ensure it reflects real-world practice,” said Professor Niek Klazinga, Em. Professor of Social Medicine, Amsterdam University Medical Centre / University of Amsterdam.
“The result is a tool that combines scientific rigour with practical usability, enabling healthcare teams to translate resilience concepts into concrete action.”
What the RESIL-Card tool is and how it works
The RESIL-Card tool is a practical online self-assessment instrument designed for use by a multistakeholder resilience team led by cardiovascular care providers.
Through a structured four-step process, including a questionnaire and guided analysis, users assess the preparedness and resilience of their cardiovascular care pathways and gain a clear understanding of how well their services can maintain care continuity during periods of disruption.
The assessment process helps teams identify existing strengths as well as potential gaps in service delivery.
Based on the responses provided, the tool offers tailored recommendations and examples of best practices to support improvement.
These insights can then inform strategic planning, helping organisations prioritise actions that reinforce care continuity, strengthen patient safety, and optimise the long-term sustainability of cardiovascular services.
Benefits for Key Stakeholders
For healthcare professionals and organisations delivering cardiovascular care, the RESIL-Card tool provides a structured way to strengthen preparedness and crisis-response capacity.
By helping teams assess their existing systems and identify areas for improvement, the tool supports better coordination across services and clinical disciplines.
It also facilitates evidence-based planning and quality improvement initiatives, enabling healthcare organisations to enhance their operational resilience while maintaining efficient and manageable care processes.
“By promoting awareness about strengths and limitations of each system, the RESIL-Card tool will help physicians to understand where improvements are needed and strengthen coordination and planning to face crises,” said Doctor Alfredo Marchese, Chief of Interventional Cardiology Department at Santa Maria Hospital, Bari, Italy and President of the Italian Society of Interventional Cardiology (GISE).
For patients and patient organisations, the RESIL-Card tool contributes to improving the reliability and continuity of essential cardiovascular care.
By encouraging healthcare providers to proactively address vulnerabilities in care pathways, the tool helps promote uninterrupted access to diagnosis, treatment, and follow-up services.
It also supports a more patient-centred and equitable approach to care delivery, encouraging collaboration and transparency in preparedness planning.
Ultimately, these improvements can contribute to better health outcomes and increased safety for people living with cardiovascular disease.
“For people living with cardiovascular disease, continuity of care is not optional — it is essential,” said Teresa Glynn, Senior Executive Strategy & Partnerships at Global Heart Hub.
“By helping healthcare providers strengthen preparedness, RESIL-Card supports more reliable and equitable access to treatment and greater confidence for patients and their families.”
At the European level, the RESIL-Card initiative contributes to a shared effort to strengthen the resilience of health systems.
By providing a common framework for assessing and improving preparedness, the tool encourages cross-border learning and facilitates the exchange of best practices among healthcare providers and policymakers.
It also aligns closely with European Union priorities on health system preparedness, crisis response, and sustainability.
By helping healthcare organisations identify vulnerabilities and implement practical resilience measures, the RESIL-Card tool can support efforts to reduce inequalities in access to high-quality cardiovascular care across EU Member States.
“Strengthening the resilience of cardiovascular care is a shared European priority,” said Rachel Kenna, Ireland’s Chief Nursing Officer at the Department of Health.
“While the RESIL-Card tool has not yet been tested in an Irish setting we look forward to seeing how it can support the development of more sustainable and prepared healthcare systems.”
Call to Action
Cardiovascular care providers and other healthcare professionals are encouraged to explore the RESIL-Card tool at https://www.wecareabouthearts.org/resil-card/online-tool/.
By using it to assess their cardiovascular care pathways, they will identify areas where resilience can be strengthened and ensure that essential services remain accessible during times of disruption.
Patient organisations also play an important role in this effort. By engaging with healthcare providers and policymakers, they can help promote the use of the tool and ensure that patient perspectives are meaningfully incorporated into preparedness and response planning.
Policymakers and health authorities are invited to support the adoption of the RESIL-Card tool within regional, national and European strategies aimed at strengthening healthcare system resilience.
Integrating the tool into policy frameworks can help safeguard access to essential cardiovascular services and enhance the ability of health systems to respond effectively to future challenges.
Learn more about Women As One at womenasone.org

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
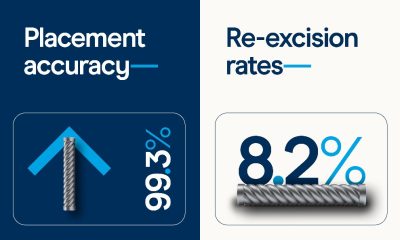
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 News4 weeks ago
News4 weeks agoResistance training has preventative effects in menopause, study finds

 Hormonal health4 weeks ago
Hormonal health4 weeks agoRelaunched women’s health strategy aims to tackle ‘medical misogyny’





























