Hormonal health
Interview: New horizons in endometriosis diagnosis
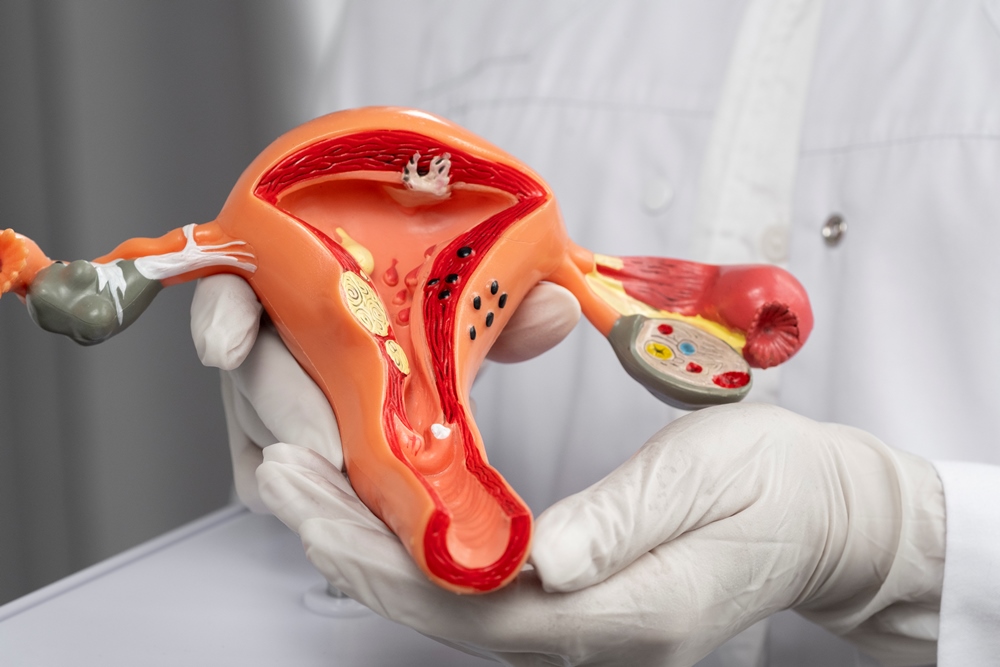
Winner of the Femtech World Menstrual Health Innovation Award 2025, Serac Healthcare, is aiming to revolutionise endometriosis diagnosis via a non-invasive diagnostic molecular imaging agent. Serac Healthcare chief executive, David Hail, speaks to FemTech World about the company’s mission to improve diagnostic timelines for the condition and empower women’s health.
Endometriosis affects more than 176 million women across the globe, causing chronic pelvic pain, painful menstrual cycles, painful intercourse, bloating and fertility issues.
Unfortunately, women are often waiting up to eight years to be diagnosed, often experiencing dismissal over their symptoms.
While scans such as MRI can provide insights into whether or not a person has endometriosis, currently, official diagnosis of the condition is done through an invasive surgical procedure called a laparoscopy, where a camera is inserted into the abdomen.
With no innovation in endometriosis diagnosis or treatment, Serac Healthcare is working to redefine how we diagnose the condition through its innovative non-invasive method.
Serac Healthcare chairman, David Hail, tells Femtech World that Serac’s imaging agent has the potential to revolutionise diagnosis and empower the development of new drugs for the condition.
“We have a real excitement and a real passion about bringing precision medicine – nuclear medicine – to people,” says Hail.
“Nuclear medicine is a way of looking at physiological processes in the body, and is very often used to see active disease in the bodies of patients, which is different to other imaging techniques.”
Serac’s imaging agent – Tc-maraciclatide – which has been granted Fast Track Designation by the FDA, is a radio-labeled tracer that has been designed to bind to new blood vessel formation.
“That’s really important in a lot of inflammatory conditions,” explains Hail.
For instance, with endometriosis, where cells from the endometrium grow outside the uterus, to be able to survive in that environment, they have to grow their own blood supply.
“That’s what we image. So, maraciclatide binds to this new blood vessel formation, which is called angiogenesis, and we can scan somebody, pinpoint that and see it.
“That is also why it works in inflammatory arthritis – rheumatoid arthritis and psoriatic arthritis, which, incidentally, is also a disease predominantly of women. In fact, autoimmune diseases generally are more prevalent in women than men, but for rheumatoid arthritis, 70 per cent of patients with rheumatoid arthritis are women.”
Hail highlights that one in two women who go for fertility treatment have endometriosis, and that these women often discover they have endometriosis when they start fertility treatment.
“At the moment, the only definitive diagnostic test for endometriosis is surgery. We know there’s a several month recovery period from laparoscopic surgery.
“In laparoscopic surgery, the surgeon can see the endometrial tissue and it is also a treatment – they can also take out the tissue, so having a definitive imaging agent that can visualize and diagnose the early stage of endometriosis is incredibly important.
“It can change the whole care pathway for some women today. When there are eight years between a woman seeing a clinician and having a formal diagnosis – apart from the physiological damage – there is psychological damage, because quite often, women are told it’s just in their heads.”
Serac has carried out clinical studies on the imaging agent, with findings from the Phase II study, carried out by a team from Oxford University, demonstrating that it can visualise and diagnose the very earliest stage of endometriosis.
“The very earliest stage of endometriosis is superficial peritoneal endometriosis (SPE) in which there are very small lesions,” explains Hail.
“Visualising this stage is really important because it is the start of the active disease, and therefore, there is the potential to intervene sooner.”
“Apart from the physiological damage and the psychological damage – there is real economic harm to the women involved. This happens at exactly the time when they are arguably at their most productive – in the early stages of their career and relationships. All these things that are negatively impacted by endometriosis.
“Our mission is about being able to bring something to the world that can make an impact on that.”
Beyond early diagnosis of the condition, Hail says that the agent may also contribute to research and the development of new drugs to treat the condition.
“This is also really important for developing new disease modifying drugs – drugs that actually get to the underlying disease, as opposed to masking them,” says Hail.
“We know that the treatments available today are not perfect – such as hormones, an example of which would be the contraceptive pill which mimics pregnancy. We know there are potential side effects from these hormonal therapies, as indeed there are from the ‘pill’.
“There are people trying to develop new disease modifying drugs, but to prove a drug works in endometriosis, you have to choose pain as an endpoint.
“There’s lots of variables that can affect pain and there isn’t a direct understanding of the relationship between the endometriosis itself and pain. The only alternative to prove that your new drug actually works and actually reduces the burden of endometrial lesions is surgery – in fact, two blocks of surgery.
“It is very difficult to run clinical trials around that for all sorts of reasons. So potentially, having this imaging agent is an alternative way to prove that your drug is actually doing what you hope it will do.”
The outline for a Phase III study has now been agreed, and Hail says that the agent being granted fast track designation in the US highlights the FDA’s recognition of the need for innovation in endometriosis care.
“Women’s Health generally, but endometriosis in particular, is an underserved area,” says Hail.
“We think it is so important to bring something to the world that’s going to make a real difference to 50 per cent of the population of the planet – we really need to do something about it, and that’s what we’re trying to do.
“Early diagnosis of endometriosis is important because it allows physicians to provide timely, effective treatment, and the best treatments that are available.”
The Femtech World Menstrual Health Innovation Award 2025 is sponsored by Clue. See all our winners here.
News
Relaunched women’s health strategy aims to tackle ‘medical misogyny’
Menopause
Watchdog bans five ads for women’s heath claims
Menopause
Non-hormonal menopause pill approved for NHS use

A new daily menopause pill approved for NHS use could bring relief to women with debilitating hot flushes and night sweats.
Around 500,000 women are expected to be eligible for the treatment, which experts say could help those unable to take hormone replacement therapy, or HRT.
The drug, fezolinetant, also known as Veoza, is a daily non-hormonal tablet designed to target the brain signals that trigger some of the most disruptive menopause symptoms.
In final draft guidance published today, the National Institute for Health and Care Excellence recommended the 45mg tablet for women experiencing moderate to severe hot flushes and night sweats.
More than two million women in the UK are thought to suffer these symptoms during menopause, often beginning during the earlier stage known as perimenopause.
For many, the effects are severe, disrupting sleep, affecting concentration and straining relationships. In some cases women are even forced to cut back on work.
An estimated 60,000 women in the UK are currently out of work or on long-term sick leave due to severe menopause symptoms, costing the economy roughly £1.5bn a year.
Research also suggests one in 10 women has left the workforce entirely because of a lack of support.

 Entrepreneur2 weeks ago
Entrepreneur2 weeks agoThree sessions that show exactly where women’s health is heading in 2026

 Menopause4 weeks ago
Menopause4 weeks agoCalifornia plans US$3.4m menopause care overhaul

 Pregnancy3 weeks ago
Pregnancy3 weeks agoHow NIPT has evolved and what AI NIPT means in 2026

 Menopause3 weeks ago
Menopause3 weeks agoWatchdog bans five ads for women’s heath claims

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoWHIS USA 2026 announces first ticket release for landmark Women’s Health Innovation Summit

 Menopause4 weeks ago
Menopause4 weeks agoMenopause has no lasting impact on cognition, research finds

 News2 weeks ago
News2 weeks agoTwo weeks left to make your mark in women’s cardiovascular health

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoQ1 momentum: Female founders are advancing, but the system still hasn’t caught up