News
Ending the guesswork in cancer care: A CEO’s vision for predictive precision oncology

By Wolfgang Hackl M.D., Founder & CEO, OncoGenomX Inc., Switzerland
During my career as an oncologist and cancer drug developer, one question haunted me more than any other: Why do some patients respond to therapy — while others, with the same diagnosis, do not?
We have innovative treatments, biomarkers, advanced lab tests, and guidelines. But far too often, despite all that science, our treatment decisions come down to educated guesswork.
As medicine evolves, so do our data — but not always our ability to act on it with precision.
That enduring gap between what we know and what we can predict inspired me to found OncoGenomX, and to build PredictionStar™, a platform designed to redefine what precision oncology truly means.
From Biomarkers to Behavior: The Missing Link
Today, most molecular cancer tests focus on eligibility: they tell us whether a tumour expresses a particular target or carries a known mutation. This is useful, but it’s only half the story.

Eligibility does not equal efficacy.
Knowing that a patient’s tumour expresses the estrogen receptor (ER), or harbours a PIK3CA mutation, doesn’t mean it will respond to hormone therapy or PI3K inhibition. It simply means those drugs might work. And in oncology, “might” is not enough.
PredictionStar™ was built to close this precision gap — by answering not just which drugs can be used, but which will actually work.
We call this Precision Drug–Tumor Matching: the ability to segregate effective from ineffective treatments by connecting genomic and phenotypic insights into a coherent tumour profile predictive of therapeutic response.
Introducing PredictionStar™: Coherent Biomarker Intelligence
PredictionStar™ is a multidimensional tumour profiling and decision-support system powered by what we term Generative Clinical Intelligence™ — the synthesis of high-quality sequencing data and AI-driven interpretation into clear, actionable clinical guidance.
 Traditional assays analyse biomarkers in isolation, treating each gene mutation or expression pattern as a separate clue.
Traditional assays analyse biomarkers in isolation, treating each gene mutation or expression pattern as a separate clue.
PredictionStar™ instead identifies logically connected biomarker constellations — genomic enablers that reveal which response mechanisms are active, and phenotypic differentiators how likely the tumour will respond.
This networked approach replaces fragmented snapshots with an integrated, functional map of tumour behaviour.
It provides oncologists with something they rarely get from today’s tests: confidence. In clinical modelling, PredictionStar™ has the potential to reduce overtreatment fivefold and lower the cost of achieving one year of tumour growth control by 35 per cent.
But the numbers tell only part of the story. Behind them are patients spared from unnecessary toxicity — and doctors empowered to treat with precision instead of probability.
Built on the Technology of Giants
PredictionStar™ was designed for seamless integration into modern real-world workflows, harmonized and cross-validated to ensure reliability, and reproducibility.

The platform’s pre-sequencing tumor workup is fully standardised, minimizing inter-laboratory variability that can otherwise reach 70 per cent.
From tumour processing to data interpretation, PredictionStar™ enforces the same rigorous quality in every step, producing consistent and concordant results across labs.
As far as cloud architecture optimized for medical data privacy and global scalability we are privileged to work with world class-players of the health IOT industry
(F. Gaede, Oct 2025, Nordcloud).
A Femtech Focus: Personalising Breast Cancer Therapy
While PredictionStar™ has broad oncology applications, our first focus is hormone receptor-positive breast cancer, the most prevalent form among women.
It is here that the limits of current diagnostics are most evident — and the need for predictive and prescriptive clarity is greatest.
 Even within hormone-dependent breast cancer, the most favorable form of the disease, patient outcomes vary widely. Some women respond beautifully to endocrine therapy for years, while others progress rapidly.
Even within hormone-dependent breast cancer, the most favorable form of the disease, patient outcomes vary widely. Some women respond beautifully to endocrine therapy for years, while others progress rapidly.
What makes the difference? The answers are buried in the tumour’s individual response profiles — but until now, we lacked the tools to decode them. PredictionStar™ offers that decoding ability.
Our non-interventional validation study, conducted in collaboration with clinical researchers from the Veterans Affairs Medical Centers in Cincinnati, Los Angeles, and Miami, involves data from over 4,300 patients with hormone receptor-positive disease.
By correlating predicted responses with actual treatment outcomes, we aim to establish a new clinical standard for predictive accuracy.
Our roadmap includes RUO and LDT certification in 2026, FDA-IDE clearance in Q2 2027, first RUO test sales as early as Q1 2027, and clinical study use from Q3 2027 onwards.
Redefining Precision Oncology
To understand why this matters, we need to reframe what “precision” means.
Most tests today are prognostic or eligibility-based. They classify risk or confirm target presence. PredictionStar™ adds a third, transformative dimension: functional prediction. It asks, “Which therapies will this specific tumour respond to — and how strongly?”
This evolution turns diagnostics into a true decision-support tool, enabling oncologists to design treatment compositions optimized for efficacy, rather than constrained by averages.
The distinction may seem subtle, but its impact for individuals living with breast cancer is enormous: Prognostic and eligibility tests describe. PredictionStar™ guides.
Innovation Through Unity
Our strength lies in collaboration.
I’ve often said that OncoGenomX stands “on the technology of giants, powered by the ambition to transform.” That is more than a slogan — it’s our reality. We built PredictionStar™ not as an isolated product, but as a platform for partnership.
Its architecture invites integration — with hospital systems, sequencing providers, AI developers, and pharmaceutical R&D pipelines.
In the coming years, we envision PredictionStar™ evolving into a broader family of tools: PredictionStar DX™ for predictive diagnostics, PredictionStar GCI™ for data integration and generation of actionable clinical intelligence, and PredictionStar IOT™ for real-time connectivity. Each module serves the same purpose: to transform complexity into clarity.
From Data to Decisions: A Personal Reflection
At its heart, PredictionStar™ was born from empathy.
As a clinician, I saw too many patients fall through the cracks — not because we lacked treatments, but because we lacked foresight.
Data without interpretation is noise. Our mission is to turn that noise into understanding.
When I speak with oncologists today, I sense both excitement and relief: “We will no longer be limited to maybes.” “We can begin to quantify response likelihood, combine therapies more rationally, and give patients something we cannot give today: certainty”.
Technology can be transformative, but only when anchored in purpose. For OncoGenomX, that purpose is simple — to give every patient the best possible chance at lasting response.
The Road Ahead
Our journey is just beginning. We are validating, scaling, and expanding across cancer types — from breast to prostate, lung, and beyond.
But our guiding principle remains unchanged: wherever there is cancer, there is a need for precision drug–tumour matching.
The convergence of genomics, phenomics, AI, and clinical data is redefining healthcare.
PredictionStar™ is part of that transformation — proving that predictive precision is not a futuristic concept, but an attainable standard.
We owe it to patients, to clinicians, and to science itself to make that standard universal.
In Closing
When I founded OncoGenomX, I imagined a world where no cancer patient has to live with uncertainty — where treatment is guided by prediction, not probability.
Today, that world feels within reach. PredictionStar™ is more than technology. It’s a promise:
That every patient deserves clarity. That every tumour can be understood.
And that, together, we can end the guesswork in cancer care.
Contact: Dr. Wolfgang Hackl | Founder & CEO, OncoGenomX | E-Mail | LinkedIn WH | Company Webpage | LinkedIn OGX
Insight
Women’s mental health happens between appointments
By Ritika Sukhani – Psychologist, part of Véa’s Clinical Advisory Board
Women’s mental health often unfolds before, between and around appointments, long before it can be neatly explained in one conversation.
A woman arrives at a GP appointment with six months of feeling “off” behind her. Not acutely unwell. Not necessarily in crisis. Just not herself.
Poor sleep. Brain fog. Lower tolerance. Irritability before her period. Anxiety before meetings. A sense that her cycle, workload, relationships and energy are interacting – but not in a way she can neatly explain when the appointment begins.
She has tracked symptoms, Googled at midnight, screenshotted articles, made notes in her phone and tried to remember what changed, when it changed and what might have triggered it.
But when she finally gets the chance to talk, the task is not only to describe how she feels. It is to organise months of fluctuating experience into a story clear enough to communicate.
This is one of the most overlooked forms of labour in women’s mental health: not simply experiencing distress but having to make it coherent.
In the NHS, women’s mental health rarely arrives through one door. It may appear in primary care as exhaustion, poor sleep or “not feeling like myself”.
It may appear in NHS Talking Therapies as anxiety or low mood. It may sit beneath a menopause conversation, a fertility journey, chronic pain, trauma, caring responsibilities, burnout or the quiet work of holding everyone else together.
The NHS provides essential care.
NHS Talking Therapies received 1.81 million referrals in 2024/25, with 1.21 million referrals accessing services and 50.5 per cent of referrals moving to recovery after completing treatment.
But women’s lives do not unfold in pathway-shaped ways.
Their distress is often cumulative, contextual and relational. It is shaped not only by symptoms but by the conditions around those symptoms: work, debt, trauma, hormones, caregiving, discrimination, physical health, relationships, sleep and the pressure to keep functioning.
That is why women’s mental health needs more than access to services.
It needs continuity.
The work before the appointment
Women are often encouraged to advocate for themselves in healthcare. At its best, this is empowering. It supports agency, preparation and active participation in care.
But self-advocacy can also become another unpaid job.
It requires women to notice what is happening, remember when it started, connect it to context, decide what feels relevant, find the right words and communicate it clearly – often while already tired, anxious, in pain or emotionally overwhelmed.
In clinical care, distress is often the beginning of an assessment. Clinicians are trained to explore duration, severity, functioning, risk, context, history and meaning.
That process matters. It is how distress becomes understood with care.
The issue is broader than any single consultation.
Women’s mental health experiences often unfold over weeks, months and years, while healthcare conversations may happen in short, pressured windows.
Patterns form outside the appointment room: between cycle phases, work demands, caring responsibilities, relational stress, poor sleep and moments of emotional overload.
The UK Government’s Women’s Health Strategy survey found that 84 per cent of respondents said there had been times when they, or the woman they had in mind, were not listened to by healthcare professionals.
It points to something structural: the difficulty of making complex, fluctuating and context-dependent experiences legible inside systems that are often fragmented, time-limited and under pressure.
Endometriosis shows why continuity, language and recognition matter.
Research has found that delays in diagnosis can occur at both patient and medical levels, including when symptoms are normalised by women themselves and by doctors.
For many women, receiving a diagnosis did more than name pain; it provided language, reassurance and possible management strategies.
This is the point we should pay attention to.
Women do not always lack information. Often, they have fragments of it.
The challenge is how to preserve context, recognise recurrence and bring scattered experiences into a form that supports reflection, communication and earlier action.
Tracking captures moments. Continuity reveals patterns
We have made enormous progress in helping women track their bodies.
We can now log cycles, sleep, mood, fertility windows, temperature, symptoms, recovery and heart rate variability with increasing precision. Period-tracking apps, for example, have been described as tools that can support body awareness and menstrual health literacy, while also introducing new forms of work, distress and privacy concern for some users.
That tension matters.
Tracking can help women notice what is happening. But noticing is not the same as understanding. And understanding is not the same as being able to explain.
A cycle tracker can show when a period started. A wearable can show disrupted sleep. A symptom log can hold isolated data points. A notes app can capture fragments of a difficult week.
But unless those pieces are brought together, the interpretive work still falls to the woman.
She is left asking: Is this hormonal? Is this stress? Is this burnout? Is this anxiety? Is this normal for me? Is this pattern important? Should I mention it? How do I explain it?
This is where the first wave begins to meet its limits.
The first wave helped women capture signals.
The next wave could help women interpret context.
The promise of patient-generated health data has always been that it could bridge the gap between everyday life and formal care. But reviews continue to highlight challenges around integration with electronic health records, trust, provenance, data quality and contextual information.
That matters because women’s health data does not become useful simply because it is collected. It becomes useful when it is contextualised.
A poor night’s sleep means something different after one stressful day than after six weeks of overextension.
A low mood entry means something different when it appears in isolation than when it appears repeatedly around a cycle phase, a work pattern or a relationship dynamic.
A spike in anxiety means something different when it is viewed alongside workload, recovery, conflict, hormonal change or self-critical thinking.
The value is not in turning every experience into a metric.
The value is in seeing what repeats, how it repeats and what else is happening around it.
Women’s mental health is systemic
The latest Adult Psychiatric Morbidity Survey found that one in five adults in England had a common mental health condition, with prevalence higher in women at 24.2 per cent compared with 15.4 per cent in men.
It also found a clear socioeconomic gradient, with common mental health conditions more prevalent in the most deprived areas and among people with problem debt.
These figures matter because they remind us that women’s mental health cannot be understood only at the level of individual coping.
Of course, individual support matters. Therapy matters. Medication may matter. Assessment, formulation, risk management and diagnosis all have their place.
But a systemic lens asks what else is happening around the woman.
Who is she caring for? What is she carrying at work? What has she had to normalise? What physical symptoms have been separated from her emotional life? What inequalities shape how quickly she is heard, referred or supported? What happens while she is waiting?
Women’s mental health is often partly biological, partly psychological, partly relational, partly occupational and partly social.
It may not fit neatly into one symptom box at first. It may need time, pattern and context before it becomes clear what kind of support is needed.
This is why the missing layer is not more monitoring – it is supported sense-making.
The need for supported sense-making
Supported sense-making means moving beyond isolated logs and dashboards towards tools that help women understand how different parts of their lives interact over time: body, mood, cycle, stress, relationships, work, recovery and self-talk.
It also means being clear about what technology should and should not do.
Technology should not replace clinical assessment. It should not diagnose from a journal entry.
It should not turn ordinary emotion into pathology or place yet another responsibility on women to optimise themselves.
At its best, it can support the wider ecosystem around care.
It can help women hold onto context before an appointment.
It can help them notice patterns while they are waiting. It can support reflection between sessions. It can help them arrive at conversations with more continuity, while respecting the clinical judgement, formulation and relational care that services provide.
This is where platforms like Véa point toward a thoughtful role for women’s health technology.
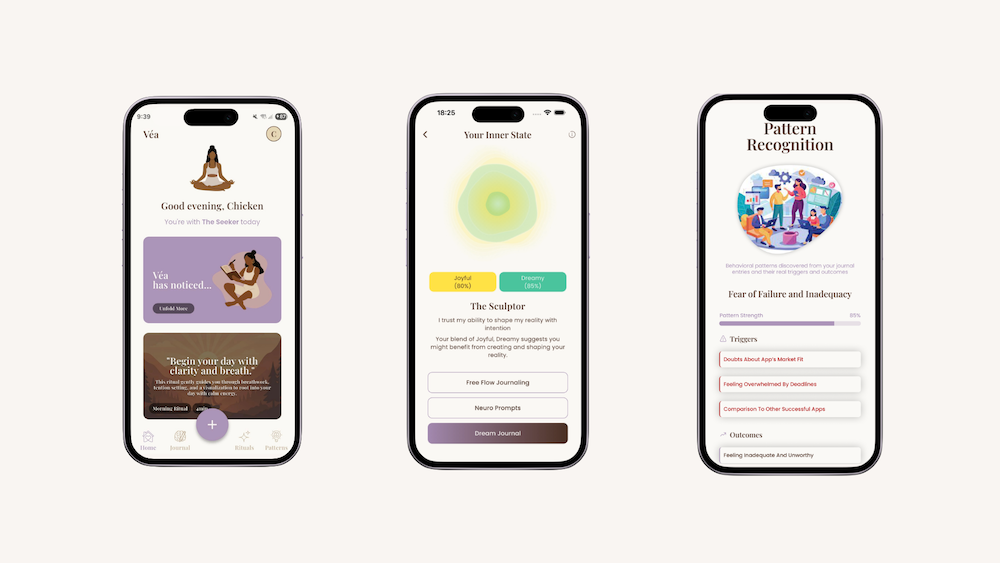
By treating reflection, emotion and language as part of women’s longitudinal health context, Véa supports the kind of pattern recognition that often gets lost in daily life. Through micro-check-ins, journalling and reflective prompts, it helps women notice shifts in their internal state over time – not to self-diagnose but to build a clearer relationship with their own patterns.
For some women, that may support a better conversation with a clinician.
For others, it may help them recognise early signs of burnout, understand cyclical changes or notice when work, relationships and recovery are interacting in ways they had not previously named.
At its best, this kind of technology reduced the burden of self-interpretation. It helps women hold onto the thread.
From more data to better continuity
The women’s health gap is often discussed as a research gap, a funding gap and a diagnostic gap.
It is all of those things. McKinsey Health Institute and the World Economic Forum estimate that closing the women’s health gap could add at least $1 trillion annually to the global economy by 2040.6
But there is another gap sitting underneath the others.
A continuity gap.
Women’s mental health experiences are often long, fluctuating and context-dependent. Healthcare systems often encounter them in snapshots. Workplaces may only notice them when performance drops. Women themselves may only recognise the pattern once they are already exhausted.
That gap between lived experience and later explanation is where too much meaning gets lost.
The next generation of women’s health technology should help preserve the story behind the symptom.
It should make room for emotional, cognitive, hormonal and social context without collapsing everything into diagnosis or optimisation. It should support better conversations without pretending to replace clinical care.
Women should not have to rely on memory alone to make sense of months of fluctuating emotional, cognitive and physical experience.
The future of women’s mental health will not be built by asking women to monitor themselves more closely.
It will be built by helping them understand themselves sooner – and arrive at conversations about their health with more continuity, context and clarity.
Learn more about Véa at veajournal.app
Cancer
Life-prolonging ovarian cancer drug approved for use in England
Diagnosis
Being female not a universal stroke risk factor for patients with AF, study finds

Female sex may not raise stroke risk across all atrial fibrillation (AF) patients, with higher risk mainly seen in women aged 75 and older, a study suggests.
Researchers said stroke prevention for women with the condition should be more personalised, especially for patients under 75.
Dr Amitabh C Pandey, director of cardiovascular translational research at Tulane University School of Medicine, said: “For years, female sex has been included as a risk factor along with other factors such as high blood pressure and diabetes, meaning women were more likely to be prescribed anticoagulants.
“Our study shows younger women may not have as much added stroke risk as previously thought, while older women, particularly those over 75, appear to have a higher risk that deserves close attention.”
The new Tulane University study challenges a long-standing assumption in heart care that being female automatically increases stroke risk for patients with atrial fibrillation.
Atrial fibrillation, often called AF, is a common heart rhythm disorder that causes the heart to beat irregularly.
It is associated with a higher risk of stroke and is often treated with anticoagulants, also known as blood thinners.
The study found that stroke risk did not increase equally across all female patients with AF.
Instead, researchers said being female may act more as a risk modifier, with increased stroke risk seen primarily among women aged 75 and older or those with a greater burden of other health conditions.
Clinicians often use a scoring system to decide whether people with AF should be prescribed blood thinners.
The system gives points for factors including age, heart failure, diabetes, previous stroke, vascular disease and high blood pressure.
Women also receive one point for sex alone.
Researchers said this can mean women with AF become eligible for blood thinners earlier or more often than men with otherwise similar risk profiles.
While blood thinners can help prevent clot-related strokes, they can also increase the risk of bruising, prolonged bleeding, gastrointestinal bleeding and other serious complications.
The researchers analysed approximately 950,000 patients with AF using TriNetX, a large anonymised electronic health record database.
They compared stroke outcomes between male and female patients across three age groups: younger than 65, 65 to 74, and 75 and older.
Male and female patients were matched based on age, other health problems and whether they had been prescribed anticoagulation medicine.
Among patients younger than 75, the study found no significant difference in one-year stroke risk between men and women.
However, among patients aged 75 and older, women had a modest but statistically significant increase in stroke risk compared with men.
In patients aged 75 and older with no additional risk factors beyond age, women had about one additional stroke per 629 patients compared with their male counterparts.
The findings support growing interest in a newer AF risk score, known as CHA2DS2-VA, which removes sex as a standalone risk factor.
However, researchers said more studies are needed and medical guidance remains inconsistent.
Han Feng, assistant professor at Tulane University School of Medicine, said: “This general approach came from women being underrepresented in AFib trials and studies comprising only about one-third of study populations.
“Our study shows not all women with AFib have the same risk profile, and these decisions should be individualised.
Pandey said: “These findings highlight the need for modern tools and approaches that can personalise risk profiles to individuals.
“The goal is not to undertreat patients who need stroke prevention, but to better identify who is most likely to benefit from anticoagulation and who may be exposed to unnecessary risk.”

 Hormonal health1 week ago
Hormonal health1 week agoPerimenopause misinformation ‘putting women at risk’

 News4 weeks ago
News4 weeks agoNIH Grant terminations disproportionately impact minority scientists, research finds

 Adolescent health4 weeks ago
Adolescent health4 weeks agoWUKA brings Period-Positive Pool Party to London Aquatics Centre to keep girls swimming through puberty

 Insight3 weeks ago
Insight3 weeks agoPCOS renamed after decade-long campaign to end ‘cyst’ misconception

 Hormonal health2 weeks ago
Hormonal health2 weeks agoNHS urged to update website following renaming of PCOS

 Menopause4 weeks ago
Menopause4 weeks agoCBT shows promise for menopause insomnia and hot flashes

 Entrepreneur1 week ago
Entrepreneur1 week agoWomen’s Health Innovation Summit opens submissions for 2026 Innovation Showcase

 News7 days ago
News7 days agoThree menopause innovators shortlisted for Femtech World Award





























1 Comment