Pregnancy
Pregnant women undertested for STIs, study finds

One in three women who tested positive for chlamydia or gonorrhoea while pregnant were not retested before giving birth, despite the potential to transmit infections to newborns.
A new study by researchers from Quest Diagnostics and the University of Alabama suggests adherence to guideline-based laboratory testing and treatment of pregnant women for two of the most prevalent sexually transmitted infections (STIs) is suboptimal in the US, with potentially dire effects.
The study, published in the Journal of Lower Genital Tract Disease, was based on deidentified results of lab tests performed in all 50 states and the District of Columbia for more than four million pregnancies.
The findings suggest gaps in guideline-based care, intended to reduce the risk of infection and medical complications.
Untreated chlamydia and gonorrhoea can raise risks of infertility and pelvic inflammatory disease in women. The risk of transmission during birth is approximately 50 per cent, raising the potential for newborns to develop infections of the eye, lungs and other health problems.
“Our study adds to a troubling body of evidence highlighting inconsistent quality in maternal and child healthcare in the US,” said Damian P. Alagia, co-author of the study and medical director of women’s health at Quest Diagnostics.
“Our analysis shows that improved adherence to existing recommendations, harmonisation of guidelines across agencies, and even updated recommendations for STI testing will be critical to ensuring the mothers and babies in the United States receive medically appropriate testing and treatment.”
Key findings:
- Over four per cent of women who received guideline-based screening for chlamydia or gonorrhoea in pregnancy during the first trimester received a positive result for one or both infections.
- More than one in three of these women (35.1 per cent chlamydia; 36.9 per cent gonorrhoea) did not receive a follow up negative test before delivery, suggesting they may not have been treated and cured (or were treated, cured but then reinfected) before birth.
- About two per cent of patients who received a negative test result for chlamydia or gonorrhoea early in pregnancy later received a positive result, suggesting “an ongoing risk of infection during pregnancy”. Of these, about one in two were still positive prior to delivery.
The authors believe reinfections (or ineffectively treated initial infections) may result in an increased number of deliveries while women are positive with one or more STIs, which can impact both maternal and newborn health.
The authors conclude current guidelines are inconsistent and they provide several recommendations for improvements.
For instance, the CDC recommends women be retested for cure at four weeks of pregnancy while the USPSTF recommends retesting before three weeks.
Current guidelines also do not recommend screening women after the age of 25 years unless there are risk factors, such as multiple partners, based on a study from 1998 when women married at younger ages than today. Guidelines recommend that all women under the age of 25 be screened, regardless of perceived risk.
The study’s strengths include its large size, national representation, and use of objective laboratory data. Its limitation is the lack of clinical follow-up information for the positive cases, as testing was limited to one national clinical laboratory.
While the authors did not evaluate other STIs, such as syphilis, they caution these patterns of irregular adherence to guideline-based testing in maternal care may extend to other conditions.
To receive the Femtech World newsletter, sign up here.
Pregnancy
App tracks heart risk after high-risk pregnancies

A recent study developed a new “digital companion” to support the prevention and follow-up of maternal cardiovascular risk in women with pregnancy complications.
Cardiovascular disease, or CVD, is the leading cause of premature death and illness in women, yet sex-specific causes remain understudied and women are underrepresented in research.
Pregnancy complications, including hypertensive disorders of pregnancy, or HDP, and gestational diabetes mellitus, or GDM, are strong predictors of future CVD, with pregnancy itself acting as a natural stress test.
Despite CVD accounting for 35 per cent of female deaths worldwide in 2019, systematic postpartum prevention remains limited in practice and incidence continues to rise.
Myocardial infarction, commonly known as heart attack, and stroke are the main fatal CVD events in women. Up to one-third of women develop hypertension within a decade after HDP, especially as maternal age rises.
Obstetric guidelines have historically lacked clarity on early CVD prevention after HDP and GDM, often relying on expert consensus rather than evidence.
Some cardiology guidelines now recommend personalised approaches, such as periodic hypertension and diabetes screening. Norwegian guidelines recommend cardiovascular risk evaluation at three months and one year postpartum, but adherence in practice is uncertain.
Effective risk reduction requires intervention before middle age. The immediate postpartum period following HDP or GDM is a critical window for early detection and intervention, offering an opportunity to engage women in cardiovascular health management, particularly as pregnancy can encourage long-term lifestyle awareness.
Electronic health, or eHealth, refers to the use of digital technologies and electronic communication tools to support healthcare services, medical information management and related health activities.
Systematic, eHealth-supported postpartum prevention can improve maternal health literacy and long-term cardiovascular outcomes.
However, there is a significant gap in targeted, eHealth-based postpartum interventions for cardiovascular risk management after HDP and GDM, despite strong patient demand and international calls for coordinated digital health strategies.
Home blood pressure monitoring shows promise, but broader digital support remains limited.
A cardiovascular postpartum follow-up programme was created as a mobile app based on Norwegian and international guidelines.
The MumCare app was developed through co-creation involving users, stakeholders and clinical experts. Five qualitative interviews and 10 user testing sessions informed improvements.
This study primarily analysed the iterative co-creation process used to develop the app, rather than evaluating clinical outcomes.
The MumCare project team in Oslo included an IT expert, obstetricians, a midwife, a GP, two sociologists and two cardiologists, all with relevant experience in eHealth and women’s health. A medical student with technological and medical expertise also helped turn ideas into app features for young women.
User representatives from two national patient associations contributed to information, recruitment, design and testing of the MumCare app.
Both associations provided user perspectives and took part in interviews and app testing. Additional users with HDP or GDM at Oslo University Hospital were also involved throughout the co-creation process.
The app’s digital infrastructure prioritises security and privacy, using encryption, de-identification and two-factor authentication.
User data is stored securely on the app and, for research purposes and with consent, on a dedicated University of Oslo server in line with GDPR and Norwegian regulations.
A linear Stage-Gate model structured the co-creation process, dividing it into phases with quality checkpoints reviewed in project meetings.
This approach balanced internal development with external user feedback, helping ensure the app is evidence-based, technically robust and user-centred.
The MumCare app guides postpartum women through tracking blood pressure, weight, physical activity and lab results, and provides personalised feedback to support self-management, mainly during the first postpartum year.
It also includes educational resources such as videos and guideline-based information to support understanding and engagement.
The app is also designed to support the transition from specialist pregnancy care to long-term follow-up with general practitioners.
It is described as a “digital companion” or health coach and does not replace clinical diagnosis or function as a medical device.
The co-creation process followed four phases focused on technical and procedural development.
In phase 1, input from expert organisations and user representatives established the app’s technical foundation.
It also reminds users of the one-year postpartum follow-up with their GP, a key time to assess risk factors and future care needs.
User organisation representatives gave feedback in phase 1, directly guiding content and feature development.
Phase 2 interviews confirmed that users want to monitor cardiovascular risk factors after HDP and GDM.
The analysis highlighted three themes: self-care strategies and uncertainties about hypertension, the need for accessible health information, and a more personalised approach to blood pressure monitoring in the app.
Concerns were also raised that frequent monitoring or app use could increase stress or create a sense of burden.
In phase 3, the app’s design and features were revised in response to feedback to improve usability and make sure they met users’ needs.
These changes led to a more intuitive and supportive interface for women during and after pregnancy.
Phase 4 involved building a prototype based on the updated designs, followed by further refinements after testing by the project team and users. Initial pilot testing with a small number of users suggested the app met its objectives and functioned as intended.
The MumCare app was co-created with input from experts, user organisations and patients over four phases.
Early expert and organisational contributions helped define the app’s goals, while ongoing feedback from patients helped ensure the design and content reflected users’ real needs.
This collaborative approach resulted in an app tailored to support women with pregnancy complications.
The MumCare app is currently being evaluated in a randomised controlled clinical trial that began in June 2024, with results needed to determine whether it improves long-term cardiovascular outcomes.
Fertility
First patients dosed in miscarriage trial
Pregnancy
Women share stories rare pregnancy complication in awareness campaign

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 Entrepreneur2 weeks ago
Entrepreneur2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
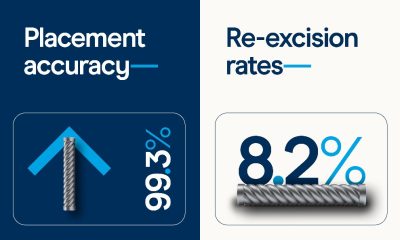
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both