News
Osteoporosis screening tool secures FDA breakthrough designation
In the US alone, 50 per cent of women over the age of 50 will experience a fracture caused by osteoporosis

London-based health tech company Naitive Technologies has received breakthrough device designation from the US Food and Drug Administration (FDA) for its osteoporosis screening tool.
OsteoSight aims to address the unmet need for earlier diagnosis of osteoporosis, a condition characterised by a progressive decrease in bone density, which is often diagnosed when a patient suffers a debilitating fragility fracture.
Worldwide, a fragility fracture caused by osteoporosis occurs every three seconds, costing global healthcare systems US$400bn annually.
In the US alone, 50 per cent of women and 25 per cent of men over the age of 50 will experience a fracture caused by osteoporosis.
The Bone Health and Osteoporosis Foundation reports that fragility fractures are responsible for more hospitalisations than stroke, breast cancer, and heart attacks combined. Despite this, it is estimated that up to 75 per cent of those affected remain undiagnosed and untreated.
Menopause significantly speeds bone loss and increases the risk of osteoporosis in women. During the menopausal transition period, the drop of oestrogen, a hormone essential for healthy bones, leads to a rapid decrease in bone density, resulting in osteoporosis.
OsteoSight aims to enhance detection rates by using the signals contained within X-rays that are taken every day, often for unrelated concerns.
By including an estimate of bone density, along with an osteoporosis classification based on World Health Organization (WHO) guidelines, into the radiology report, Naitive says its technology could alert physicians sooner to their patient’s bone health.
“While treatments exist that can help slow the progression of bone loss, the biggest challenge we face is the persistent underdiagnosis of osteoporosis and low bone density,” explained Robert Pignolo, Professor of Medicine, Geriatric Medicine and Gerontology at the Mayo Clinic and member of the scientific advisory board of Naitive Technologies.
“Patients are still being diagnosed after a fracture, instead of during a period of timely intervention. By screening patients who are having routine X-rays, we have a real opportunity to identify early bone loss and osteoporosis and prevent fractures.
“I’m delighted that the FDA has seen OsteoSight’s potential, and I’m excited to see the difference it will make in clinical practice.”
The FDA’s Breakthrough Device Program is intended to provide patients with more timely access to medical devices that have the potential for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
As part of the programme, the FDA will expedite development, assessment, and review of OsteoSight for regulatory clearance.
Will Briggs, founder and CEO of Naitive, said: “We are thrilled that OsteoSight has received Breakthrough Device Designation.
“It’s a key milestone for our technology and validates our strong belief that OsteoSight has the potential to revolutionise the way we find patients at-risk for osteoporosis.
“We look forward to bringing this technology to market and helping improve patient outcomes.”
News
Perimenopause may offer “window of opportunity” for heart disease prevention
News
Research project of the year shortlist revealed

The Femtech World Awards is proud to reveal the shortlist for Research Project of the Year as part of the third annual global celebration of innovation, impact and leadership across women’s health.
From fertility science and perimenopause research to regional ecosystem analysis, the shortlisted projects reflect the breadth and growing influence of femtech research worldwide.
The category is sponsored by OncoGenomX, with the winner to be selected by a representative from the organisation.
OncoGenomX is dedicated to offering solutions and providing comprehensive support services that empower Drug Developers, Clinical Researchers, Oncologists,NextGenSeq Diagnostics Laboratories, NextGenSeq Service Organisations, Cancer Diagnostics and Therapeutics Companies to achieve their ambitious goals
The shortlisted entries for Research Project of the Year are:

Women’s health remains significantly underserved in South-East Asia, with persistent gaps in access, awareness, and quality of care carrying substantial social and economic costs.
This report examines the femtech landscape in Indonesia, the Philippines, Singapore, Thailand, and Vietnam, highlighting market trends, emerging technologies including artificial intelligence, and the evolving support ecosystem.
It identifies key challenges facing femtech founders, including limited access to finance, low awareness and persistent stigma, marketing constraints linked to content moderation, and gaps in tailored ecosystem support.

Led by Stephanie Willson, MD, of the IVI RMA Global Research Alliance, the study explored whether embryos that show certain chromosome abnormalities during genetic testing may still have the potential to result in a healthy pregnancy and live birth.
The research analysed more than 7,600 frozen embryo transfers and found that some embryos previously considered unlikely to succeed were still capable of leading to successful pregnancies, although at lower rates than embryos without abnormalities.
The findings could help fertility clinics and patients make more informed decisions during IVF treatment, particularly in cases where there are limited embryos available.
Rather than automatically discarding these embryos, the research supports a more evidence-based and personalised approach to fertility care.

For many women, perimenopause can feel confusing and unpredictable, with limited research explaining what is happening in their bodies.
Natural Cycles set out to change that by leading one of the largest studies ever conducted on menstrual and ovulatory patterns, uncovering new insights into how ovulation behaves as women approach menopause.
Conducted in collaboration with researchers from George Washington University, Seattle Clinical Research Center, Gennev and the University of California San Diego, the study analysed nearly one million menstrual cycles from more than 197,000 women aged 18–52 across more than 140 countries.
The scale of this dataset made it possible to explore menstrual patterns and ovulation in far greater detail than has traditionally been possible in women’s health research.
The Femtech World Awards celebrates the innovators, researchers and organisations driving meaningful progress in women’s health.
What happens next
Winners across all categories will be revealed during the virtual ceremony on June 19, with winners receiving a trophy and an interview with a Femtech World journalist.
Mental health
Women over 40 seeking raves for mental health benefits

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
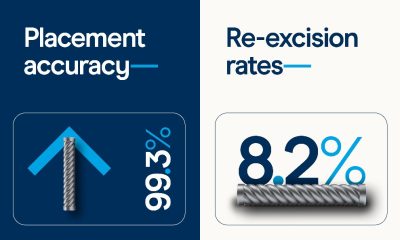
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Menopause3 weeks ago
Menopause3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both





























