Menopause
Osteoporosis screening tool secures FDA breakthrough designation
In the US alone, 50 per cent of women over the age of 50 will experience a fracture caused by osteoporosis

London-based health tech company Naitive Technologies has received breakthrough device designation from the US Food and Drug Administration (FDA) for its osteoporosis screening tool.
OsteoSight aims to address the unmet need for earlier diagnosis of osteoporosis, a condition characterised by a progressive decrease in bone density, which is often diagnosed when a patient suffers a debilitating fragility fracture.
Worldwide, a fragility fracture caused by osteoporosis occurs every three seconds, costing global healthcare systems US$400bn annually.
In the US alone, 50 per cent of women and 25 per cent of men over the age of 50 will experience a fracture caused by osteoporosis.
The Bone Health and Osteoporosis Foundation reports that fragility fractures are responsible for more hospitalisations than stroke, breast cancer, and heart attacks combined. Despite this, it is estimated that up to 75 per cent of those affected remain undiagnosed and untreated.
Menopause significantly speeds bone loss and increases the risk of osteoporosis in women. During the menopausal transition period, the drop of oestrogen, a hormone essential for healthy bones, leads to a rapid decrease in bone density, resulting in osteoporosis.
OsteoSight aims to enhance detection rates by using the signals contained within X-rays that are taken every day, often for unrelated concerns.
By including an estimate of bone density, along with an osteoporosis classification based on World Health Organization (WHO) guidelines, into the radiology report, Naitive says its technology could alert physicians sooner to their patient’s bone health.
“While treatments exist that can help slow the progression of bone loss, the biggest challenge we face is the persistent underdiagnosis of osteoporosis and low bone density,” explained Robert Pignolo, Professor of Medicine, Geriatric Medicine and Gerontology at the Mayo Clinic and member of the scientific advisory board of Naitive Technologies.
“Patients are still being diagnosed after a fracture, instead of during a period of timely intervention. By screening patients who are having routine X-rays, we have a real opportunity to identify early bone loss and osteoporosis and prevent fractures.
“I’m delighted that the FDA has seen OsteoSight’s potential, and I’m excited to see the difference it will make in clinical practice.”
The FDA’s Breakthrough Device Program is intended to provide patients with more timely access to medical devices that have the potential for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
As part of the programme, the FDA will expedite development, assessment, and review of OsteoSight for regulatory clearance.
Will Briggs, founder and CEO of Naitive, said: “We are thrilled that OsteoSight has received Breakthrough Device Designation.
“It’s a key milestone for our technology and validates our strong belief that OsteoSight has the potential to revolutionise the way we find patients at-risk for osteoporosis.
“We look forward to bringing this technology to market and helping improve patient outcomes.”
Menopause
Enter the menopause innovation award before it’s too late

If you are working in menopause care and have not yet entered the Femtech World Awards, you have until this Friday, 17 April, to put your work forward.
The award celebrates those leading the way in reshaping how menopause is understood and supported across healthcare and society.
The winner will have demonstrated exceptional innovation in addressing the health, wellbeing and quality of life needs of people going through this transition.
Judges will assess impact, inclusivity, accessibility and the ability to challenge stigma while delivering meaningful, real-world solutions.
The scope is intentionally broad.
Whether you have developed a digital platform, a diagnostic tool, a pharmaceutical or non-pharmaceutical treatment, a workplace support programme or something that does not sit neatly within a single category, if your work is improving the menopause experience, this award is for you.
Who is behind the award
The category is sponsored by Cross-Border Impact Ventures (CBIV), an impact venture capital firm investing in early growth stage health technology companies across medical devices, diagnostics, therapeutics and digital health.
Every company CBIV supports must show relevance to women’s, children’s and adolescents’ health, with the ambition to scale into emerging markets.
Annie Thériault, managing partner at Cross-Border Impact Ventures, said: “Being part of the FemTech World Awards gives us a front-row seat to the most exciting breakthroughs in women’s health.
“It’s a powerful way to stay connected to the pulse of innovation and the future of care.”
What you stand to gain
Entry is free.
Every shortlisted organisation receives extensive coverage across all Femtech World platforms, placing your innovation in front of a global audience of investors, clinicians, industry leaders and potential partners.
The winner also receives a trophy and a dedicated interview.
The deadline is this Friday
Nominations and entries close on 17 April.
After that, the Femtech World team will shortlist the strongest submissions, with the final decision made by a representative from CBIV.
Find out more about the awards and enter for free here.
Menopause
Premature menopause raises long-term heart risk by 40%, study finds

Women who enter natural menopause before age 40 face about a 40 per cent higher lifetime risk of developing coronary heart disease than women who experience menopause later, according to a large study that is the first to calculate lifetime heart risk associated with premature menopause.
The findings suggest that doctors should routinely ask women about age at menopause, using the menopausal transition as an opportunity to identify higher-risk women and intervene earlier.
Dr Priya Freaney is assistant professor of medicine in the division of cardiology at Northwestern University Feinberg School of Medicine.
She said: “When menopause happens before age 40, women still have more than half of their life expectancy ahead of them.
“Understanding their cumulative lifetime risk of blockage-related heart disease is critical.”
Coronary heart disease is a condition where the heart’s arteries become blocked or narrowed by a buildup of fatty deposits called plaque.
By restricting blood flow to the heart, these plaques can lead to sudden events (heart attacks) or gradual damage (weakened heart muscle).
The study of more than 10,000 U.S. women followed for decades also found that premature menopause was three times more common among Black women than white women (15.5 per cent vs. 4.8 per cent).
According to Freaney, the disparity likely reflects a complex mix of life-course exposures, health conditions and structural inequities rather than solely inherent biological differences.
Freaney and colleagues analysed data from 10,036 postmenopausal Black and white women who participated in six long-running U.S. studies, including the Framingham Heart Study, Atherosclerosis Risk in Communities Study and the Women’s Health Initiative.
The women were followed between 1964 and 2018.
During that time, the Northwestern scientists found more than 1,000 cases of coronary heart disease events in the data, including fatal and non-fatal heart attacks.
Even after accounting for cardiovascular risk factors such as smoking, obesity, hypertension and diabetes, premature menopause was associated with 41 per cent higher risk of coronary heart disease for Black women and 39 per cent increased risk for white women.
The scientists note in the study that the causes of premature menopause are not fully understood and are likely multifactorial.
Potential contributors include genetic, biological and environmental factors, as well as earlier age of the first menstrual period, health behaviors (such as smoking), obesity and the cumulative effects of chronic stress.
It is also unclear whether the menopausal transition itself creates a vascular environment that promotes disease, or whether women who experience premature menopause already have an underlying risk profile that predisposes them to both premature menopause and cardiovascular disease.
Even at the average age, menopause’s hormonal changes can affect cardiovascular health.
During menopause, declining estrogen levels trigger changes that increase coronary heart disease risk.
“As the natural estrogen declines, no matter what age it happens in, cholesterol and blood pressure go up, body fat distribution shifts to the abdomen, muscle mass gets lower, blood sugars can become dysregulated and arteries stiffen,” said Freaney, who also is director of the Women’s Heart Care Program at Northwestern Medicine Bluhm Cardiovascular Institute.
“Together, these changes over a short period increase the risk of heart disease.”
Freaney said women who experience premature menopause should think of it as an early signal to take their heart health seriously.
“Tell yourself: I have to be far more proactive than my neighbor about my own heart health,” Freaney said.
“The vast majority of heart disease is preventable, but people need to know that they’re at risk early in life because effective prevention takes decades.
“Tell your doctor, ‘I experienced premature menopause. What can we do to protect my heart?’” she suggests.
The findings also highlight a gap in how menopause is discussed in medical care, according to Freaney.
“All clinicians need to get comfortable asking about menopause because we have estrogen receptors from our head to our toes.”
For years, menopause has largely been treated as a gynecologic issue, she said. But the hormonal transition affects nearly every system in the body, including the cardiovascular system.
That means cardiovascular clinicians should routinely ask about menopause history when assessing long-term cardiovascular risk.
“Historically, women have been vastly understudied in cardiovascular science, and we still have much to learn about how menopause influences heart health,” Freaney said.
Menopause
Cardiff opens its first women’s health hub as nationwide rollout begins

Cardiff’s first women’s health hub has opened, offering specialist perimenopause and menopause support for women aged 40 to 65.
Minister for mental health and wellbeing Sarah Murphy visited the East Cardiff Menopause Hub this week to learn about the new service, which brings together patient-centred advice, treatment and community support under one roof.
The hub forms part of a Wales-wide network of pathfinder women’s health hubs established during the first phase of delivering the Women’s Health Plan.
Women registered with one of the East Cardiff GP Cluster practices will be able to access extended 20-minute consultations, available face-to-face, by telephone or online, with GPs who have specialist experience in menopause care.
Practice nurses will also provide broader healthcare support, including blood pressure checks, lifestyle advice and guidance on hormone replacement therapy.
A Menopause Café, open to women of all ages, will offer a welcoming community space to share experiences and ask questions.
Sarah Murphy said: “It was fantastic to visit the Maelfa hub and see first-hand how Cardiff and Vale University Health Board is bringing high-quality, compassionate care closer to home for women in East Cardiff.
“Women’s health hubs will make it easier for women in Wales to get care when they need it.
“As the pathfinder hubs are rolled out, we’ll be listening to women’s feedback and adapting to make sure we are building a health service which meets the needs of women and girls, now and for generations to come.”
By March, every health board in Wales will have a pathfinder women’s health hub. Each health board has received an additional £300,000 this financial year to support their development.
The hubs form part of the Women’s Health Plan, which includes more than 60 actions to close the gender health gap and is based on feedback from around 4,000 women across Wales.
Dr Claire Beynon, executive director of public health at Cardiff and Vale University Health Board, said: “Too many women feel unsupported or unheard when seeking help for the symptoms of menopause.
“The East Cardiff Menopause Hub is a really positive step in bringing high-quality, compassionate care closer to home, with longer appointments and specialist expertise focused on women’s health needs.
“By combining clinical care with community support, this service helps women feel informed, confident and in control of their health. It also reflects our wider commitment to reducing health inequalities.”

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
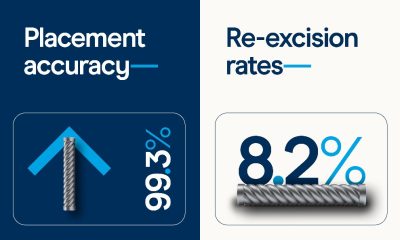
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both




























