To receive the Femtech World newsletter, sign up here.
News
Contraception app raises US$55m to ‘fill gaps’ in women’s health
Natural Cycles is the only digital form of birth control on the market cleared by regulators around the world

The contraception app Natural Cycles has secured US$55m in funding to “fill the gaps” in underserved areas of women’s health.
Natural Cycles is the first direct-to-consumer contraceptive app that aims to help women plan or prevent a pregnancy “naturally” through an algorithm that analyses daily hormone-driven temperature changes to confirm if the user is fertile.
The app remains the only digital form of birth control on the market cleared by regulators in the US, Europe, Canada, Australia, Singapore and South Korea.
The US$55m funding round, led by Lauxera Capital Partners with participation from Point72 Private Investments and a revolving debt facility from J.P. Morgan, is hoped to help the Stockholm-based company invest in commercial acceleration, product development and healthcare reimbursement automation.
“We’ve experienced profitable growth over the last few years as women actively seek more birth control options and we’re excited to welcome the next phase of Natural Cycles journey together with Lauxera Capital Partners and Point72 Private Investments,” said Dr Raoul Scherwitzl, co-founder and CEO of Natural Cycles.
“We look forward to leveraging their expertise to fulfil Natural Cycles’ mission of making hormone-free birth control more accessible and combining technology and science to fill the gaps in underserved areas of women’s health.”
Samuel Levy, founding partner of Lauxera Capital Partners, added: “Our mission at Lauxera is to partner with ambitious entrepreneurs transforming the future of medicine with unique technology.
“The visionary Natural Cycles team has built an exceptional company delivering profitable growth supported by unique clinical and regulatory business moats.
“We are delighted to partner with Elina, Raoul and their team to offer women an alternative to traditional approaches without the side effects and compromises.”
As part of the investment, Levy will join Natural Cycles’ board of directors.
Lauxera Capital Partners and Point72 Private Investments join Natural Cycles’ current investors, including EQT Ventures, Samsung Ventures, Heartcore Capital, Headline and Bonnier Ventures.
Pregnancy
Early birth safer in high blood pressure pregnancies – study
News
Low insulin diet and avoiding four food groups may prevent menopause weight gain
News
Three brain and mental health innovators shortlisted for award

We are excited to reveal the three exceptional innovators shortlisted for the Brain & Mental Health Innovation Award at the Femtech World Awards 2026.
The award recognises groundbreaking work addressing the cognitive and emotional health challenges that uniquely and disproportionately affect women.
This award is sponsored by Women In Cloud, the global network of 120,000 women tech founders, executives, professionals and allies across 80 countries, united in their mission to make the tech ecosystem an inclusive force for change – and to unlock US$1 billion in new economic access by 2030.
The shortlisted entries will now be judged by a representative from Women In Cloud who will announce the winner at a virtual event on June 19.
Congratulations to the shortlist and thank you to everyone who entered or nominated.
Brain and Mental Health Innovation Shortlist

HealCycle is a clinical and social breakthrough targeting what founder Ananya Grover calls the “Silent Decade” – the years when women’s endocrine health and environmental stressors are routinely dismissed by traditional healthcare silos.
Under the clinical leadership of psychiatrist Dr. Aninda Sidhana, HealCycle monitors HPO-axis markers, addresses conditions like PMDD, and integrates an AI companion, Tara, built on the principle of Radical Empathy.
Backed by the WICCI National Women’s Mental Health Council and designed to meet WHO standards for gender-responsive care, HealCycle is replacing silence with science – from Delhi to the world.

Môr is reimagining cognitive wellbeing from the ground up with the first science-led, female-first nutritional system designed around how women’s brains actually work.
Grounded in compelling research – including findings from Weill Cornell Medicine showing accelerated metabolic brain decline during menopause transition – Môr’s patented AM/PM chrono-targeted architecture delivers the right ingredients at the right time: daytime mental clarity and stress resilience in the morning, nervous system recovery and sleep support in the evening.
With backing from Innovate UK and a clinical feasibility study in development, Môr is building for the invisible majority: cognitively depleted women who have been failed by a market that never built for them.

Véa is an AI-powered emotional operating system that builds a living cognitive profile of each user, helping women understand their thoughts, triggers and behavioural patterns through neuroscience-backed journalling.
Véa does not just record how women feel, it connects the dots across weeks, months and years to reveal the hidden architecture of their emotional world.
Having already analysed over 101,000 anonymised words journaled to identify 3,000+ specific cognitive distortions – such as ‘double-bind’ guilt and ‘catastrophising’ – Véa is closing the critical gender data gap in mental health.

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
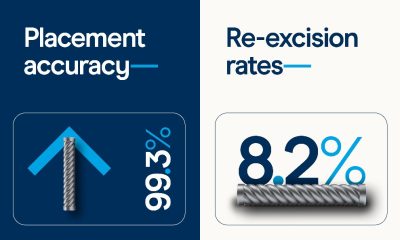
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy4 weeks ago
Pregnancy4 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now

 Wellness3 weeks ago
Wellness3 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage

 Opinion2 weeks ago
Opinion2 weeks agoWhat Maternal Mental Health Month reveals about where postpartum support actually breaks down

 Fertility3 weeks ago
Fertility3 weeks agoToxins and climate harms having ‘alarming’ effect on fertility, research warns




























7 Comments