News
Telehealth abortion service expands across the US to allow people to have safe abortions at home
Remote services help people have an abortion where the medical procedure remains legal post Roe v Wade

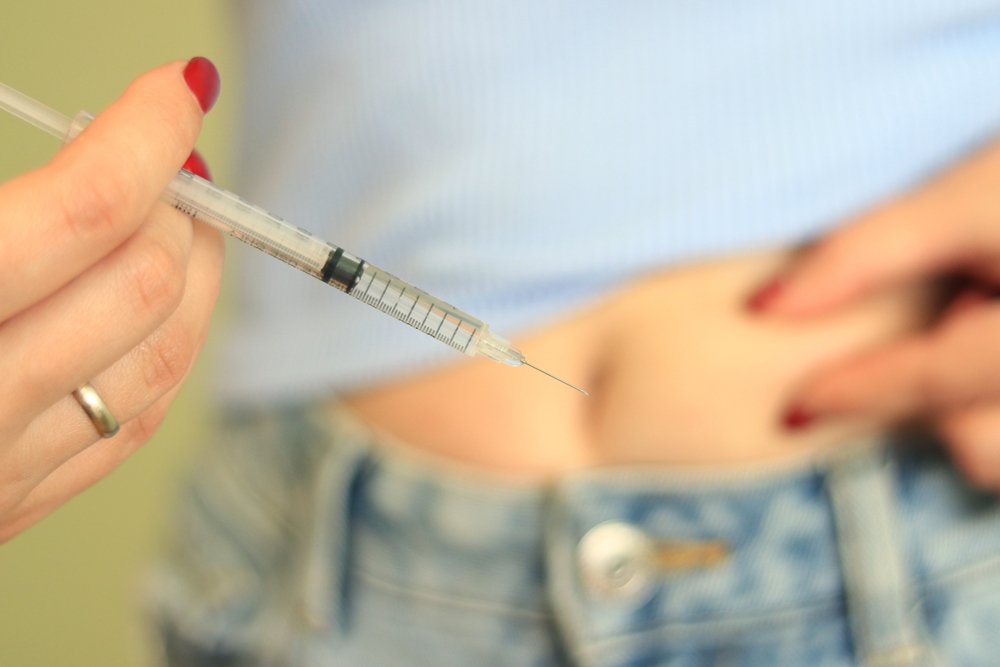
A telehealth medical abortion service expands its offering in nine more states to allow people to complete safe abortions at home.
Wisp first launched abortion care in August in California and has now launched its telehealth services in Connecticut, New Mexico, Colorado, Illinois, Maryland, Maine, New York and Washington.
The sexual and reproductive health provider has became now the largest, most accessible medical abortion provider in the US.
The company offers abortion pills via telehealth because of new rules introduced by the Food and Drug Administration (FDA) during the pandemic.
Prior to that, people had to see providers in person in order to get mifepristone, one of the two drugs used in a medical abortion.
However, the agency relaxed the rules early in the pandemic and lifted them last year.
With a shipping time of one to two business days, the telehealth provider offers services estimated to be 30 per cent less expensive than the national average cost of medical abortion. The treatment consists of oral medication to terminate a pregnancy.
Following the Supreme Court’s decision to overturn Roe v Wade, some states have banned telehealth services for abortions.
Services like Wisp help expand abortion access in places where the medical procedure is already legal.
“In the wake of the overturn of Roe v Wade, inequities in access to care have only deepened,” said Ahmad Bani, CEO of the company.
“By providing the most accessible abortion services in more states than ever, we’re able to help alleviate a stressed healthcare system and allow patients to take ownership of their reproductive health with privacy and respect.”
The new launch expands Wisp’s suite of specialised services for sexual and reproductive health, from emergency contraception and birth control to bacterial vaginosis and herpes.
Fertility
Researcher explores weight loss jab impact on PCOS
Diagnosis
Researchers teach AI to spot cancer risk by squeezing individual breast cells
Diagnosis
Experimental drug drowns triple-negative breast cancer cells in toxic fats

An experimental drug slowed triple-negative breast cancer in mice by flooding tumour cells with toxic fats.
Triple-negative breast cancer lacks three common drug targets, making it one of the hardest-to-treat and most aggressive forms of the disease.
The compound, known as DH20931, appears to push cancer cells past their limits by triggering a surge in ceramides, fat-like molecules that place the cells under intense stress until they self-destruct.
In lab experiments, the drug also made standard chemotherapy more effective. When combined with doxorubicin, researchers were able to reduce the dose needed to kill cancer cells by about fivefold.
The drug targets an enzyme known as CerS2 to sharply increase production of these lipids and stress cancer cells. Healthy cells, by contrast, showed lower sensitivity to the drug in lab tests.
While the early results are promising, further preclinical and clinical trials would still be needed to determine the safety and effectiveness of DH20931 in humans.
Satya Narayan, a professor in the University of Florida’s College of Medicine, led the study with an international group of collaborators.
The researchers published their results on human-derived tumours on 21 April and presented their findings on combination therapy at the annual meeting of the American Association for Cancer Research in San Diego.
Narayan likened the drug’s effects to a home’s electrical system handling a power surge.
While healthy cells act like a properly grounded and installed circuit, cancer cells are more like a jumble of mismatched wires and faulty fuses. DH20931 overwhelms cells not with electricity, but with fats.
He said: “When that surge goes into the cancer cells, they cannot handle the amount of power they are getting. The fuses burn out, the cell can’t handle the surge and it dies.”
The compound was developed at the University of Florida in the lab of Sukwong Hong.
Hong, now a professor at the Gwangju Institute of Science and Technology in South Korea, created DH20931 as one of many drug candidates tested for efficacy in Narayan’s lab.
In the study, researchers implanted human triple-negative breast cancer tumours into mice and treated them with DH20931.
The drug significantly slowed tumour growth without causing noticeable weight loss or signs of toxicity in the animals. In separate lab experiments, it also showed activity against other breast cancer subtypes.
In addition to increasing lipid levels, DH20931 triggers a second stress signal by flooding cells with calcium.
Together, these effects disrupt the mitochondria, the structures that produce a cell’s energy, ultimately leading to cell death.
Narayan said: “It does not just follow one pathway but it goes through multiple pathways. It’s a two-hit hypothesis.
“These pathways are common in all breast cancer types and other solid tumours, so we think this drug can be useful not only in triple-negative breast cancer but potentially other cancers as well.”

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoThree sessions that show exactly where women’s health is heading in 2026

 Entrepreneur4 days ago
Entrepreneur4 days agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 Pregnancy4 weeks ago
Pregnancy4 weeks agoHow NIPT has evolved and what AI NIPT means in 2026

 News4 weeks ago
News4 weeks agoTwo weeks left to make your mark in women’s cardiovascular health

 Opinion4 weeks ago
Opinion4 weeks agoQ1 momentum: Female founders are advancing, but the system still hasn’t caught up

 Fertility2 weeks ago
Fertility2 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic

 Menopause2 weeks ago
Menopause2 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health6 days ago
Mental health6 days agoLifting weights shows mental health and cognitive benefits in older women, study finds





























3 Comments