Entrepreneur
Sword Health launches bloom: a new pelvic pain product

SWORD Health has launched Bloom, a physical therapy product aimed at women struggling with pelvic pain
Bloom is a clinical-grade digital solution aimed at women’s physical health issues. It will help to provide non-invasive care for neglected health issues such as pelvic pain or postpartum recovery. Worldwide, The rates of chronic pelvic pain for women of childbearing age range from 14 per cent to 32 per cent globally.
It includes a sensor that will measure the pressure and stamina of a person’s pelvic floor while connecting to an app so that users can receive feedback on their exercises. Patients will then be paired with a pelvic health specialist who will guide them through exercises or tailor the program to suit their needs.
It is hoped that Bloom will help to reduce chronic pain and dysfunction in turn decreasing the need for painkillers or invasive options, like surgery.
Bloom and pelvic pain
Marta Cardeano, general manager of Bloom said: “I’ve suffered many years with pelvic pain, and it’s now an honour to build this solution that will help thousands of women across the world. Just like me, many women simply don’t know there are solutions that can help free them from pain.
We are here to support the female population with effective and non-invasive solutions to improve their quality of life. Bloom technology is providing a new way for women to address their pelvic disorders from the privacy and convenience of their homes.”
Virgilio Bento, CEO of SWORD Health said: “Women’s physical health has been an overlooked problem in healthcare for decades. Right now, one in every four women suffers from pelvic disorders, without being able to access a solution that is both clinically effective and convenient.
That’s why we’ve decided to launch Bloom: we believe every woman should be able to have access to the best care and not suffer in silence. After developing the new gold standard of care for back and joint pain, Bloom is our next step towards fulfilling our mission to free two billion people from pain”.
Entrepreneur
Just 24 hours left to nominate your company of the year

You have until Friday to nominate your femtech company of the year.
The award is one of 10 featuring at Femtech World’s third annual awards event, which attracts entries from across the UK, EU and Europe.
The Company of the Year Award is for companies that have demonstrated exceptional leadership in tackling women’s health needs through groundbreaking products, services or platforms that are shaping the future of global femtech.
If your company is driving innovation, impact and growth in this space, this award was made for you.
About the sponsor: Femovate
The category is backed by Femovate, the global femtech incubator using design to fuel innovation across every stage of a woman’s health journey, from proactive prevention through to personalised treatment.
Femovate has invested over US$2 million in design capital, working side-by-side with founding teams to bring market-ready solutions to life.
The startups it supports have collectively raised US$120 million, launched 30 products, and secured seven FDA clearances.
Why enter?
The Femtech World Awards are free to enter.
Winners and shortlisted companies receive extensive coverage across all Femtech World platforms.
Winners will also receive a trophy and the opportunity to be featured in an interview for the publication.
Find out more about the Femtech World Award and enter here by 4pm BST on Friday 17.
Entrepreneur
The disciplined advantage: Wellness for modern leadership

By Chaitra Vedullapalli, founder and president, Women in Cloud
Today’s leaders are carrying more than responsibilities.
They are carrying caregiving roles, financial pressures, legal complexities, and the quiet emotional weight of sustaining performance in unpredictable times.
And yet, most leadership forums still focus on productivity, growth, and scale, rarely addressing the human systems that enable sustained leadership.
At Women in Cloud, we believe the next generation of leadership will not be defined by endurance alone, but by discipline, preparation, and self-respect.
That is why we are proud to announce the #WICxWellness Summit 2026: The Disciplined Advantage.
This is a wellness summit focused on leadership preparedness.
Why This Summit Matters Now
Across our global community, a clear pattern has emerged.
- More professionals and executives are quietly navigating:
- Life-threatening health crises, personally or within their families
- Financial stress caused by medical, caregiving, or business shocks
- Legal and healthcare decisions with long-term consequences
- Career interruptions due to caregiving responsibilities
- Chronic exhaustion, grief, and emotional isolation
These experiences are widespread, yet rarely discussed in executive settings.
#WICxWellness 2026 brings these realities into the open with compassion, clarity, and practical guidance, so leaders can prepare, adapt, and continue forward without breaking.
This is wellness as a leadership discipline.
What We Will Explore
This year’s summit is designed to provide grounded, immediately applicable insights for founders, executives, and senior leaders.
Executive Function Under Chronic Stress
Keynote with Jennifer Brown

Focus Areas:
- Understand how chronic stress alters judgment, focus, and emotional regulation
- Identify early warning signs of cognitive overload in leadership roles
- Adaptability as a Cognitive Anchor: Navigating rapid change without compromising decision-making.
- Resilience Through Connection: Building human-centric teams to mitigate the stress of uncertainty.
- Navigating Change with Emotional Intelligence: Regulating stress responses to maintain clarity and empathy.
- Systemic Agility in High-Pressure Environments: Creating foundations of psychological safety that protect organizational function
Labs That Matter (And What to Do With Them)
Fireside Chat with Chelsey Galipeau and Chaitra Vedullapalli

Focus Areas:
- Which health tests are worth the time and investment
- Interpreting results without panic
- Preventive strategies for executive longevity
- Avoiding unnecessary medical spending
Caretaking Without Collapse
Panel With Karen Fassio, Anca Platon Trifan, Scilla Andreen, Clara Schroeder, Sheena Yap Chan

Focus Areas:
- Managing family and professional demands without burnout
- Setting humane boundaries
- Building sustainable support systems
- Ending silent sacrifice in leadership
Food as a Stability System (Not a Diet)
Fireside Chat with Nancy Watt and Meagan T. Copelin

Focus Areas:
- Nutrition for sustained energy and clarity
- Blood sugar and decision-making
- Realistic eating during travel and stress
- Food as operational resilience
Autoimmunity, Hormones & Cardiac Risk in Real Life
Fireside Chat with Dr. Linda Bing

Focus Areas:
- Managing invisible health risks
- Hormonal transitions and stamina
- Flare prevention at work
- Energy mapping for leadership performance
A Different Kind of Leadership Conversation
At Women in Cloud, we have learned something fundamental:
Performance is visible. Discipline is invisible.
Yet discipline determines whether leaders can serve, build, and lead over decades, not just quarters.
The Disciplined Advantage is about building that resilience intentionally.
Through science, lived experience, and practical frameworks, this summit equips leaders to:
- Anticipate strain instead of reacting to a crisis Design sustainable work rhythms
- Protect cognitive and emotional capital
- Lead from grounded strength
This is leadership infrastructure for the next decade.
Join Us
If you are a founder, executive, technologist, or senior professional navigating complexity and determined to lead well without sacrificing your health, clarity, or longevity, this summit is for you.
The #WICxWellness Summit 2026 is where performance meets preservation. Where ambition meets sustainability. Where leadership becomes truly durable.
We look forward to welcoming you. The Disciplined Advantage begins here.
Secure your spot: https://www.eventbrite.com/e/wicxwellness-summit-2026-tickets-1981616382945?aff=oddtdtcreator
Entrepreneur
Who will be crowned Startup of the Year?

Femtech World is continuing the search for a company to be crowned Startup of the Year.
The award is one of 10 to feature at the third annual Femtech World Awards.
The Startup of the Year Award celebrates an early-stage company making a bold impact in women’s health through innovation, vision and execution.
The winning startup will have demonstrated strong potential to transform care, accessibility, or awareness in women’s health with a scalable solution.
Consideration will be given to innovation, market traction, inclusivity, impact and the ability to address unmet needs.
The award is sponsored by Future Fertility.
The company is transforming fertility care with AI-powered solutions that close critical information gaps along the IVF journey.
Its clinically validated, non-invasive tools analyse oocyte images to predict each egg’s reproductive potential, supporting decision-making across key pathways: VIOLET™ for egg freezing, MAGENTA™ for IVF-ICSI, and ROSE™ for donor programmes and egg banks.
These reports deliver personalised insights into egg quality and ploidy potential, empowering patients and clinicians to make more informed decisions regarding next steps.
Today, Future Fertility’s technology is used in more than 300 clinics across 35+ countries.
Developed with the world’s largest oocyte image dataset linked to reproductive outcomes, Future Fertility’s AI models generate quality scores that not only guide treatment planning and manage expectations, but also serve as objective, actionable KPIs for labs—driving improved outcomes and transparency in fertility care worldwide.
The awards are free to enter, with winners receiving a trophy and an interview with Femtech World.
Find out more about the awards and enter for free here.

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 Entrepreneur2 weeks ago
Entrepreneur2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Fertility4 weeks ago
Fertility4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
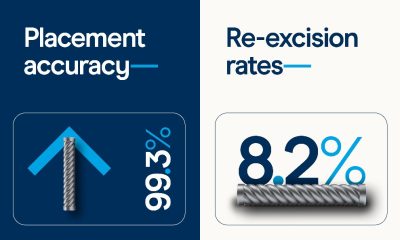
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Fertility4 weeks ago
Fertility4 weeks agoFlora Fertility closes US$5m seed round

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds


























