To receive the Femtech World newsletter, sign up here.
News
Start-up secures FDA clearance for stress urinary incontinence device
The device can be used during a specific activity or up to 12 hours for general control over symptoms

The US women’s health start-up Watkins-Conti has obtained 510(k) clearance from the US FDA for its stress urinary incontinence device.
Yōni.Fit is a device intended for the temporary management of urine leakage caused by stress urinary incontinence (SUI) in women 18 years and older.
The silicone device is a soft vaginal insert that aims to reduce urine leaks without interfering with voluntary urination. It is self-administered and can be used during a specific activity or up to 12 hours for general control over symptoms.
The FDA clearance represents a “substantial milestone” in non-surgical options for the temporary management of urinary incontinence, a condition estimated to affect about one in three women in the US at some point in their lives.
Allison L. Watkins, founder and CEO of Watkins-Conti, said: “My lived experience as a new mother with SUI gave me the inspiration for Yōni.Fit.
“The options available to me at the time did not complement my lifestyle as a working mom with two small children.
“Listening to patients on their journey and innovating accordingly is paramount. I am committed to developing even more solutions for women’s pelvic health, using the Yōni.Fit device as a self-care platform that could potentially include diagnostics and drug delivery.”
Fifty-eight participants were enrolled in a randomised, controlled study of Yōni.Fit in women with SUI. The study was conducted by urogynaecologists at Stanford, NYU Langone, and Jefferson Health.
Of the participants completing the evaluation phase, a significantly higher percentage experienced clinically meaningful reduction in 12-hour pad weights with the Yōni.Fit device compared to the control device.
Karolynn T. Echols, director and associate professor of female pelvic medicine and reconstructive surgery at Thomas Jefferson University and an investigator in the Yōni.Fit clinical study, said: “My patients with stress urinary incontinence want a solution that is effective and convenient – one that accommodates the reality of their busy lives and does not interfere with voluntary urination.
“The cost and downtime associated with surgery can be prohibitive for some women.”
Dr Kate C. Arnold, chief medical officer of Watkins-Conti, added: “I believe Yōni.Fit will be an appealing and effective self-care solution for patients who value convenience and want to be in control of their pelvic health.
“We are committed to helping all women, including those in rural areas and in medically underserved communities, where access to surgery would be challenging.”
Ross Watkins, president of Watkins-Conti, shared: “With this FDA clearance, Watkins-Conti is now positioned to help this important and underserved market.”
News
Research project of the year shortlist revealed

The Femtech World Awards is proud to reveal the shortlist for Research Project of the Year as part of the third annual global celebration of innovation, impact and leadership across women’s health.
From fertility science and perimenopause research to regional ecosystem analysis, the shortlisted projects reflect the breadth and growing influence of femtech research worldwide.
The category is sponsored by OncoGenomX, with the winner to be selected by a representative from the organisation.
OncoGenomX is dedicated to offering solutions and providing comprehensive support services that empower Drug Developers, Clinical Researchers, Oncologists,NextGenSeq Diagnostics Laboratories, NextGenSeq Service Organisations, Cancer Diagnostics and Therapeutics Companies to achieve their ambitious goals
The shortlisted entries for Research Project of the Year are:

Women’s health remains significantly underserved in South-East Asia, with persistent gaps in access, awareness, and quality of care carrying substantial social and economic costs.
This report examines the femtech landscape in Indonesia, the Philippines, Singapore, Thailand, and Vietnam, highlighting market trends, emerging technologies including artificial intelligence, and the evolving support ecosystem.
It identifies key challenges facing femtech founders, including limited access to finance, low awareness and persistent stigma, marketing constraints linked to content moderation, and gaps in tailored ecosystem support.

Led by Stephanie Willson, MD, of the IVI RMA Global Research Alliance, the study explored whether embryos that show certain chromosome abnormalities during genetic testing may still have the potential to result in a healthy pregnancy and live birth.
The research analysed more than 7,600 frozen embryo transfers and found that some embryos previously considered unlikely to succeed were still capable of leading to successful pregnancies, although at lower rates than embryos without abnormalities.
The findings could help fertility clinics and patients make more informed decisions during IVF treatment, particularly in cases where there are limited embryos available.
Rather than automatically discarding these embryos, the research supports a more evidence-based and personalised approach to fertility care.

For many women, perimenopause can feel confusing and unpredictable, with limited research explaining what is happening in their bodies.
Natural Cycles set out to change that by leading one of the largest studies ever conducted on menstrual and ovulatory patterns, uncovering new insights into how ovulation behaves as women approach menopause.
Conducted in collaboration with researchers from George Washington University, Seattle Clinical Research Center, Gennev and the University of California San Diego, the study analysed nearly one million menstrual cycles from more than 197,000 women aged 18–52 across more than 140 countries.
The scale of this dataset made it possible to explore menstrual patterns and ovulation in far greater detail than has traditionally been possible in women’s health research.
The Femtech World Awards celebrates the innovators, researchers and organisations driving meaningful progress in women’s health.
What happens next
Winners across all categories will be revealed during the virtual ceremony on June 19, with winners receiving a trophy and an interview with a Femtech World journalist.
Wellness
Women over 40 seeking raves for mental health benefits
News
Osteoporosis significantly increases risk of death in menopause, study suggests

Osteoporosis may raise the risk of death in postmenopausal women by up to 47 per cent, a new study suggests.
The findings point to an inverse relationship between femoral bone mineral density and mortality risk, especially within certain ranges.
Femoral bone mineral density is the amount of mineral in the thigh bone, which is often measured to assess bone strength and osteoporosis risk.
Dr Monica Christmas is associate medical director for The Menopause Society.
She said: “Osteoporosis often remains a silent threat after menopause, despite its profound effect on women’s lives—from loss of height, poor balance, and reduced mobility to disfigurement, pain, and even premature death.
“Early screening and preventive measures, including a calcium-rich diet (preferably from food sources), regular weight-bearing exercise, and hormone therapy when appropriate, can significantly improve bone health and reduce risks not only of fractures but also cardiovascular disease, certain cancers, and dementia.
“It’s time we bring this conversation to the forefront.”
In the study involving nearly 3,000 postmenopausal women, bone mineral density at four femoral sites was assessed using dual-energy x-ray absorptiometry, a scan commonly used to measure bone strength and fracture risk.
The analysis found that mortality risk was significantly higher when femoral bone mineral density reached the osteoporotic threshold or when osteoporotic fractures were present.
After full adjustment, osteoporosis was associated with a 47 per cent increased risk of mortality.
A stronger inverse association between increased bone mineral density and mortality risk was seen within specific ranges, suggesting bone mineral density could serve as a prognostic marker of wider health.
The relationship appeared especially notable within the range of 0.46 to 0.71 g/cm² for total femur bone mineral density.
Previous research has shown that postmenopausal women face a significantly higher risk of death within one year of hip or vertebral fractures.

 Entrepreneur3 weeks ago
Entrepreneur3 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News2 weeks ago
News2 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report

 Diagnosis4 weeks ago
Diagnosis4 weeks agoFuture Fertility partners with Japan’s leading IVF provider, Kato Ladies Clinic
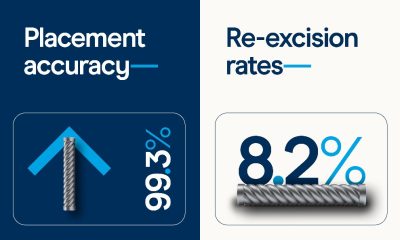
 Diagnosis3 weeks ago
Diagnosis3 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker

 Menopause4 weeks ago
Menopause4 weeks agoMore research needed to understand link between brain fog and menopause, expert says

 Mental health3 weeks ago
Mental health3 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 News3 weeks ago
News3 weeks agoResistance training has preventative effects in menopause, study finds
 Pregnancy3 weeks ago
Pregnancy3 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both



























5 Comments