News
EngagedMD raises US$11m in funding to make fertility care more accessible
The company’s software integrates patient education and engagement into existing clinical workflows to reduce clinical burden

A Washington, DC-based technology company has raised US$11m in funding to make fertility care more accessible.
The funding round, led by the venture capital firm MonCap, will help EngagedMD to expand its global presence and expansion into other healthcare verticals.
As part of the investment, EngagedMD has also welcomed Jonathan Sockol, managing partner of MonCap, to the company’s board.
The company currently supports more than half of all patients in the US, UK, and Canada through its cloud-based SaaS platform that empowers patients and providers to collaborate and make informed decisions.
Its software integrates patient education and engagement into existing clinical workflows to reduce clinical burden and improve patient experiences and outcomes.
The company says clinics using EngagedMD save up to two hours of manual time per patient, allowing them to provide better and more accessible care to more patients.
A number of medical journals have also clinically validated the firm’s impact on patient experience, noting benefits such as enhanced knowledge, control over decision making, and overall satisfaction scores.
“We are excited to partner with MonCap on EngagedMD’s next chapter of growth,” said Taylor Stein, co-founder and co-CEO of EngagedMD.
“MonCap has a proven track record of adding value and their expertise will help us further our mission of making patients’ and providers’ lives easier.
His co-founder, Jeff Issner, said the company would use the funds to continue developing products that support providers and patients navigating a complex care journey.
“We also look forward to expanding our platform into other specialties in our relentless journey to make healthcare more efficient and effective,” he added.
MonCap’s Sockol said: “EngagedMD has already positively impacted the lives of over one and a half million patients. We are proud to back this team in making better care more accessible to even more patients across healthcare.”
Diagnosis
Women unaware of gynaecological cancers

Only one per cent of women can name all five gynaecological cancers, new research suggests, as 21 women in the UK die every day of the diseases.
The report also found that 31 per cent of women have put off or avoided seeking medical advice for gynaecological symptoms.
It also found that 43 per cent of women invited for cervical screening said barriers had put them off attending, while 18 per cent of respondents aged 25 to 34 who had been invited had never attended.
The five main gynaecological cancers are womb, also called uterine, ovarian, cervical, vulval and vaginal cancer.
The Lady Garden Foundation said that, while progress has been made since the UK government’s 2022 Women’s Health Strategy aimed to improve gynaecological cancer care, significant challenges remain.
John Butler, medical director and trustee at the Lady Garden Foundation, said: “The fact that only one per cent of the population can name the diseases that directly affect half of us underscores a significant awareness gap, impacting individuals’ ability to recognise vital signs and symptoms or seek timely medical help.
“Addressing this isn’t just about awareness; it’s a critical public health priority. Our collective efforts are essential to ensure the latest commitments announced by this government translate into tangible change that saves lives.”
The report said key reasons for delaying medical advice included difficulty making appointments, embarrassment and, for cervical screening, fear of pain or previous bad experiences.
Women also reported challenges within healthcare interactions, including feeling “not taken seriously”, “dismissed” or “not believed” when seeking gynaecological advice.
Jenny Halpern Prince, chief executive and charity co-founder, said: “We frequently hear reports of women feeling ‘not taken seriously,’ ‘dismissed,’ or ‘not believed’ when seeking gynaecological advice.
“These experiences highlight crucial areas where we can improve patient support and trust within our healthcare system, ensuring women receive the empathetic and effective care they need.”
The Lady Garden Foundation said it aims to increase awareness of both the charity and the five gynaecological cancers.
It also aims to serve as a primary entry point for reliable, stigma-free information, helping people understand their bodies, recognise symptoms and overcome barriers to accessing care.
Its Silent No More Garden was unveiled at the RHS Chelsea Flower Show 2026. Designed by Darren Hawkes, the garden serves as a national call to action, using five sculptures to spark conversations, break long-standing taboos and encourage open dialogue about symptoms and preventative care.
Butler said: “Continued focus and collaborative action are essential to progress.
“The ongoing commitment from the government, alongside societal efforts to break down taboos surrounding gynaecological health, are crucial.
“The Lady Garden Foundation is dedicated to being a beacon of information and support, empowering women with the knowledge they need. We urge everyone to learn the signs, speak up, and help us save lives.”
Mental health
Poor sleep linked to Alzheimer’s risk in older women – study

Poor sleep may signal higher Alzheimer’s risk in older women with greater genetic risk, a study suggests.
Older women who reported poorer sleep also showed greater memory difficulties and more Alzheimer’s-related brain changes, the study found.
That pattern appeared only in women with higher genetic risk, suggesting sleep complaints may be a stronger warning sign for some women than for others.
Researchers examined 69 women aged 65 years and older taking part in the Women Inflammation Tau Study, an ongoing project focused on ageing and Alzheimer’s disease risk.
Participants completed questionnaires about their sleep quality, underwent memory testing and received brain scans measuring tau. Tau is a protein that accumulates abnormally in Alzheimer’s disease.
The study found that poorer self-reported sleep was associated with worse visual memory performance and greater tau accumulation in brain regions affected early in Alzheimer’s disease, but only among women with higher genetic risk.
Women with lower genetic risk did not show the same relationship between sleep complaints, memory and tau build-up. The finding was specific to visual memory and was not observed for verbal memory.
Researchers said the results add to growing evidence that sleep disturbances and Alzheimer’s disease may reinforce one another over time.
Previous studies have suggested that disrupted sleep can contribute to the build-up of abnormal tau proteins, while Alzheimer’s-related brain changes may also interfere with healthy sleep patterns.
Because women account for nearly two thirds of Alzheimer’s cases and frequently report poorer sleep quality than men, the researchers said sleep may represent an important and potentially modifiable risk factor in older women.
The authors noted that self-reported sleep assessments are inexpensive and easy to administer, raising the possibility that sleep complaints could help identify people who may benefit from closer monitoring or early intervention.
They also suggested that improving sleep could become a target for future Alzheimer’s prevention strategies, particularly for women at elevated genetic risk.
Fertility
AI could transform ovarian care through personalisation, study finds
AI could transform ovarian care by personalising cancer and fertility treatment, but more clinical validation is needed before routine use.
A systematic review and meta-analysis found AI models showed high diagnostic accuracy for ovarian cancer when combining data such as ultrasound scans and blood test results.
Across 81 studies, AI models correctly identified ovarian cancer in around nine out of 10 cases, with pooled rates of 89 to 94 per cent.
They were also highly accurate at ruling out ovarian cancer when it was not present, with specificity of 85 to 91 per cent.
The analysis also found that explainable AI tools could predict complete surgical cytoreduction in advanced ovarian cancer.
Complete surgical cytoreduction means removing all visible cancer during surgery, which can be an important goal in treatment planning.
The tools achieved a pooled AUC of 0.87. AUC is a measure of how well a model distinguishes between different outcomes, with higher scores showing stronger performance.
In reproductive medicine, AI algorithms helped physicians optimise ovarian stimulation protocols and predict follicular growth during IVF.
Ovarian stimulation is the use of hormones to encourage the ovaries to produce eggs, while follicles are the small sacs in the ovaries where eggs develop.
The review found AI could reliably model ovarian response in IVF with a pooled AUC of 0.81.
However, researchers said challenges remain in translating promising research findings into routine clinical practice.
They identified substantial variation across studies, driven by retrospective study designs, variable AI systems and a lack of standardised validation.
Only 22 per cent of analysed studies reported prospective, multicentre external validation, where models are tested forward in time across multiple healthcare settings.
The authors called for rigorous validation to help close the gap between research and routine clinical practice, alongside standardised methodological and reporting frameworks, smooth integration with clinical workflow and robust governance to support responsible and ethical AI use.
They concluded: “Artificial intelligence is a transformative force in the management of ovarian conditions.
“In gynaecologic oncology, AI enhances every phase of care, from early detection and accurate diagnosis to prognostic stratification and surgical planning.”
In reproductive medicine, AI personalises ovarian stimulation and refines the diagnosis of heterogenous endocrine disorders such as PCOS.
PCOS, or polycystic ovary syndrome, is a hormonal condition that can affect periods, skin, weight and fertility.

 Entrepreneur4 weeks ago
Entrepreneur4 weeks agoFuture Fertility raises Series A financing to scale AI tools redefining fertility care worldwide

 News3 weeks ago
News3 weeks agoWomen’s digital health market set to reach US$5.28 billion in 2026 – report
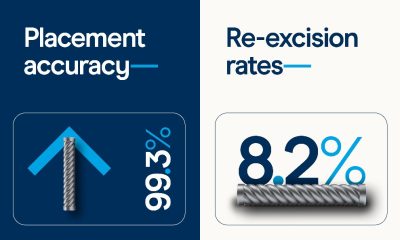
 Diagnosis4 weeks ago
Diagnosis4 weeks agoNew meta-analysis further supports low re-excisions and high placement accuracy with the Magseed marker
 Pregnancy4 weeks ago
Pregnancy4 weeks agoNIPT or NT scan? Why the 2026 evidence supports doing Both

 Mental health4 weeks ago
Mental health4 weeks agoLifting weights shows mental health and cognitive benefits in older women, study finds

 Wellness4 weeks ago
Wellness4 weeks agoResistance training has preventative effects in menopause, study finds

 Insight3 weeks ago
Insight3 weeks agoWhy the UK’s fertility rate keeps falling – and what it means if you’re trying now

 Events3 weeks ago
Events3 weeks agoWomen’s HealthX unveils Northwell Health, Corewell Health, Biogen & more to headline Chronic Disease stage
























1 Comment