News
Managers still unprepared to discuss menstrual health, study finds

Few HR professionals believe managers are properly trained to support employees with endometriosis and other menstrual health conditions, new research has revealed.
Only 16 per cent of HR professionals said line managers had the training or resources to effectively support staff with menstrual health issues, according to new research.
That figure dropped to 13 per cent for endometriosis – a disease affecting the lining of the womb, which impacts one in 10 women and those assigned female at birth from puberty to menopause.
The research, carried out by HR software provider Ciphr and the charity Endometriosis UK, surveyed 121 HR professionals and leaders in summer 2025.
It also found that many employers do not provide free period products or have flexible uniform rules.
Julie Burns is the Endometriosis Friendly Employer scheme manager at Endometriosis UK.
She said: “We know that there are many HR professionals already putting in place the right measures to enable those with endometriosis to feel valued, be productive and contribute to their organisation’s success – but as a charity we also hear stories of women and those assigned female at birth treated unfairly or without sympathy, forced to change careers or abandon their ambitions, because of the disease.
“We hope HR professionals recognise that providing support to the estimated 1.5m in the UK with endometriosis is in their interests.
“Doing so will not only enable that community to achieve their potential, but it sends a message to your wider team that they are valued and can expect support and reasonable adjustments.”
Menstrual health policies were far less common among UK employers (11 per cent) than those for mental health (57 per cent) or peri/menopause (48 per cent).
However, flexible working – offered by 89 per cent of employers – may provide some support for employees with endometriosis.
Only 21 per cent of employers recorded data on how many staff had a diagnosed or suspected menstrual health condition such as endometriosis.
Meanwhile, 29 per cent said free period products were not supplied at work, with another 3 per cent unsure.
Among organisations where uniforms are worn, just 18 per cent said employees were asked whether adjustments were needed.
This could be particularly relevant for those with endometriosis, who may experience bloating or heavy bleeding that can temporarily alter body shape by up to two dress sizes.
Claire Hawes, chief people and operations officer at Ciphr, said employers should take the findings seriously.
She said: “It’s concerning that these results reveal such a wide gap in organisational awareness and readiness to support employees living with endometriosis and other menstrual health conditions.
“When managers are perceived as unapproachable or ill-equipped to respond with empathy and understanding, organisations risk sidelining the needs of a significant part of their workforce.
“HR teams have a vital role to play in changing this. Managers must be empowered to support their people holistically – not just in terms of performance and KPIs, but in their health and wellbeing too.
“That means investing in training, implementing flexible policies, signposting resources, and fostering open, compassionate conversations about health.”
Sanchia Alasia is a trustee of Endometriosis UK and senior HR professional who lives with endometriosis.
Alasia said: “My personal and professional experience shows me that a little bit of flexibility can go a really long way.
“Those with endometriosis need to know that if they have a flare-up or need to attend a medical appointment, they can tell a manager and be believed and understood, rather than judged.
“That flexibility needs to be there both in the culture of an organisation and in the policies that line managers rely on.
“Ultimately, line managers need clarity, and all sorts of employees may need that flexibility at some time or another, regardless of whether they have endometriosis, another medical condition, or other circumstances affecting them.”
Diagnosis
Lymph nodes could reveal who’s most at risk of breast cancer spreading
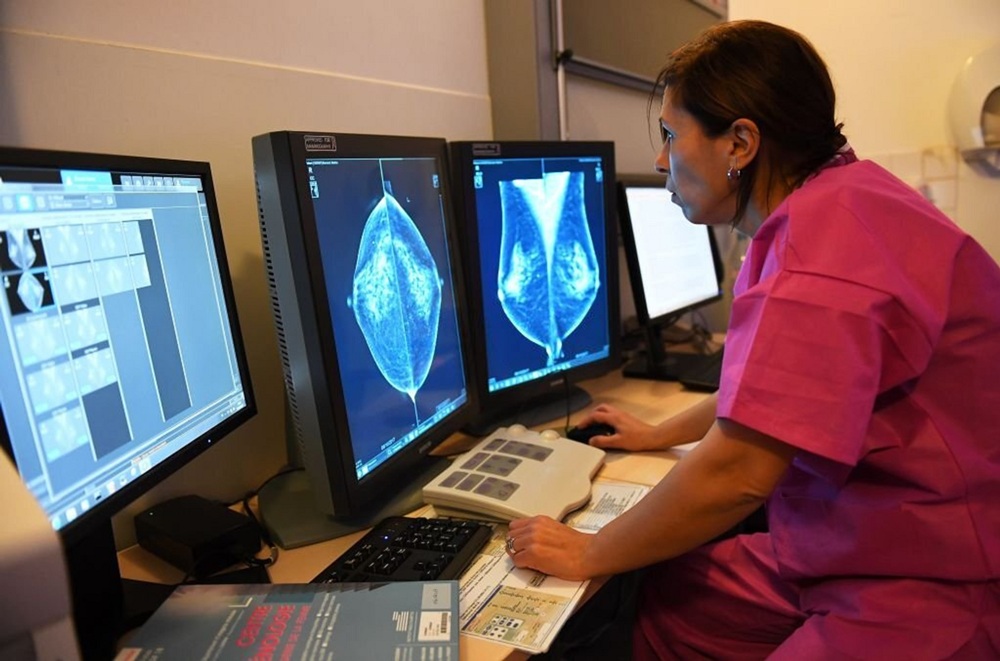
Changes in lymph nodes may help show which breast cancer patients face higher or lower risk of the disease spreading, researchers have found.
The findings could support more tailored care, new treatments and help more people avoid unnecessary treatment.
Dr Simon Vincent is chief scientific officer at Breast Cancer Now, which funded the research:
He said: “These findings suggest that changes to the structure of the lymph nodes are more than just a consequence of the cancer. They can also play an active role in helping breast cancer progress.
“With one person tragically dying from breast cancer every 45 minutes in the UK, we urgently need research like this so that we can better understand who is most at risk of their cancer progressing and becoming incurable. Only then we can find ways to stop it.
“With a better understanding of how lymph nodes change as breast cancer spreads, we could find new targets for future treatments for types of breast cancer that are harder to treat.”
Lymph nodes, a key part of the immune system, help the body fight infections and cancer. In breast cancer, the lymph nodes in the armpit are often the first place the disease spreads to.
At the moment, everyone with invasive breast cancer has to undergo surgery to remove lymph nodes so doctors can check for cancer cells.
Invasive breast cancer means cancer that has spread beyond where it first developed in the breast into nearby tissue.
While this is effective, it can lead to long-term side effects such as swelling of the arm, known as lymphoedema, and may be unnecessary for some patients, particularly those with early-stage disease or those whose cancer responds well to treatment.
The study analysed 331 lymph node samples from people with different types of breast cancer and compared them with healthy lymph nodes from people free from the disease.
It found that breast cancer could change the structure of a network that supports the lymph nodes.
Crucially, some of these changes could occur before doctors were able to spot any cancer cells in the network.
Some changes were linked to a better chance of survival, while others were associated with a poorer prognosis.
Dr Amy Llewellyn and Dr Kalnisha Naidoo from King’s College London, together with professor Sophie Acton at University College London, compared the 331 samples with healthy lymph nodes in people free from the disease.
They looked at fibroblastic reticular cells, known as FRCs, a group of cells in lymph nodes that provide their structure, control fluid flow and activate different immune cells.
The study showed that the structure of this FRC network could change before the cancer had spread and differed depending on the type of breast cancer, any spread and whether someone had received chemotherapy.
Chemotherapy uses medicines to kill cancer cells or slow their growth.
The researchers said the findings could help doctors better understand who is most at risk of breast cancer spreading.
Dr Llewellyn said the first large-scale analysis of FRC in human lymph node tissue from breast cancer patients was addressing the “urgent need” for a better understanding of the area’s biology.
News
Why proven women’s health innovations still can’t find a home

By the Health Innovation Exchange
For more than a decade, femtech’s scale gap has been treated as a funding problem. What if that diagnosis is incomplete?
Despite growing attention, women founders still receive just 2 per cent of global venture funding, and years of advocacy have failed to shift the needle.
This persistence is no longer just a concern; it signals a deeper structural failure.
This is not just a funding gap. It is a system failure.
As Pradeep Kakkitill, founder and CEO of the Health Innovation Exchange (HIEx), argues, the sector continues to operate on a flawed assumption.
The belief that better support to founders alone will unlock scale overlooks the deeper structural constraints that determine whether the innovation is adopted at all.
Barriers That Go Beyond Capital
These insights are not theoretical. Global research reinforces that these challenges are not isolated, highlighting structural, financial and systemic barriers that shape how women-led and under-represented ventures access funding, markets and pathways to scale.
Importantly, these findings are not draw from research alone, but from the lived experiences of women and under-represented founders themselves.
Across HIEx-led interviews and focus group discussions conducted as part of the Reckitt Catalyst Programme, founders repeatedly described the same challenges: fragmented financing, unclear adoption pathways, repeated cycles of proof, and systems that lacked clear routes from validation to procurement and scale.
These experiences suggest that the barriers facing women-led innovation are not simply financial. They are structural.
Many high-potential ventures are not failing because funding is absent.
They are failing because the systems that determine scale, including public procurement, regulation and financing, are not built to move proven solutions beyond pilots into widespread adoption.
This is not a founder problem. It is a system design failure.
Beneath these structural constraints sits a more persistent challenge. Entrenched attitudes shaped by unconscious bias continue to influence decision making.
Across investment and public-sector systems, innovation led by women and underrepresented founders is still frequently perceived as higher risk.
These perceptions shape how opportunities are evaluated, increase the burden of proof placed on founders, and slow decision making. In practice, this results in systematically higher barriers to both funding and adoption.
Systems Unable to Absorb Innovation
Dr. Abas Hassen, lead executive officer for health innovation and quality at Ethiopia’s Ministry of Health, underscores this point.
The primary constraints are not about innovation quality, but about the systems that determine adoption and scale, including procurement, regulation, financing and delivery.
He identifies three persistent challenges: institutional resistance to change, “pilot purgatory” where solutions are repeatedly tested but not integrated into public systems, and a disconnect between what external funders support and what governments can sustain.
Ethiopia’s response reflects a broader shift. Innovation is no longer treated as isolated pilots, but as a structured component of system design.
The country’s system-led innovation model combines regulatory pathways, prioritisation frameworks and structured testing environments to embed innovation directly within the health system.
The implication is clear.
Scaling innovation is not only about accelerating individual ventures alone. It is about strengthening the systems that determine whether innovation is adopted at scale.
The Missing Middle: From Pilot to Procurement
In many low- and middle-income countries, public systems remain the largest market for health and WASH solutions, accounting for the majority of service delivery and procurement.
Yet capital is deployed through models that do not reflect this reality, as scaling depends on public-sector adoption, long procurement cycles and regulatory integration rather than rapid returns.
This creates a misalignment within the financial ecosystem, where capital is structured for faster high returns, while impact depends on long-term system integration.
At its core, the challenge is the absence of clear adoption pathways.
Without structured routes from validation to procurement and system-wide use, even effective solutions struggle to move beyond pilots.
This is the “missing middle”, the gap between early validation and large-scale adoption.
The consequences of this “missing middle” are perhaps best illustrated by the founders trying to navigate it.
Temie Giwa-Tubosun, founder and CEO of LifeBank, describes her decade-old company as an “orphan” within existing financial structures, too commercial for impact investors and too impact driven for venture capital.
Businesses operating within health systems often fall between funding models that were not designed for them.
Thato Schermer, co-founder of Zoie Health, describes a similar challenge.
Even companies with strong revenue and clear demand struggle to secure funding at the right stage, as they are assessed through frameworks that do not reflect the healthcare markets.
Across interviews and focus group discussions, these patterns were consistent.
Founders described fragmented financing, unclear adoption pathways, and repeated cycles of proof, where they are asked to keep proving their solutions without a clear route to scale.
These are not isolated challenges. They reflect how innovation is funded, evaluated and integrated across the system.
The barrier to scale is not a lack of viable solutions. It is about the systems and models that are not designed to support them.
Reducing Risk Through System Design
From an HIEx perspective, a different approach is emerging, one that focuses not on fixing founders, but on designing how systems manage risk and adopt innovation.
Rather than avoiding risk, Ethiopia is working to manage it through structured processes.
The system is “risk-aware, not risk-averse.” It uses innovation sandboxes, structured testing environments within public systems that allow new solutions to be evaluated under controlled conditions.
These mechanisms, generate decision-grade evidence while limiting system-wide exposure, creating clearer pathways from validation to adoption.
When innovations are tested within public systems, they gain institutional legitimacy. This reduces perceived risk for both governments and investors and enables more confident decision making.
From Fragmentation to Coordination
Within this context, initiatives such as Reckitt Catalyst, a multi-partner platform supporting women-led health and WASH innovation to scale, play a critical bridging role.
By connecting entrepreneurs with governments, investors and technical partners, and aligning solutions with national priorities, the programme helps to create clearer pathways from pilot to procurement and scale.
But alignment alone is not enough.
As Pradeep Kakkattil notes, the climate movement offers a useful parallel. Climate progress was not driven by evidence alone. It accelerated when investors, governments, and institutions began treating inaction as the greater risk.
Sustained pressure exposed the cost of doing nothing, redefined how risk was assessed and ultimately reshaped capital allocation and policy decisions.
Women’s health and WASH innovation is now at a similar inflection point.
Despite years of evidence and advocacy, outcomes such as women receiving a fraction of global funding persist.
This is not due to a lack of solutions. It is because the systems governing investment, adoption and scale have not been sufficiently challenged.
What is required is not incremental progress.
It is a shift in what the system tolerates – how risk is defined, how capital is allocated, and how accountability is enforced.
A System at an Inflection Point
The implications are clear.
Investors must move beyond rigid funding models and deploy capital aligned to how health systems scale. Governments must build clearer pathways for testing, procurement and adoption.
Ecosystem actors must shift from supporting individual ventures, to enabling system-level integration.
The persistent funding gap is not a result of slow progress; it reflects a system operating exactly as designed. Incremental change will not shift outcomes.
What is required is a fundamental reset of how femtech is financed and scaled: from passive investment to active market-shaping, where capital, policy, and procurement work together to create real pathways to adoption.
Until that shift happens, the sector will continue to produce innovation that the market is not structured to absorb.
News
Menopause workplace toolkit launched to help UK employers support staff

A new free menopause toolkit has been launched to help UK employers respond better to menopause at work, improve wellbeing and retain experienced staff.
Wellbeing of Women has launched MENO-Kit in partnership with Lancaster University, which it describes as the UK’s first evidence-based menopause workplace toolkit.
The online resource translates more than a decade of academic research into practical guidance for employers.
It is designed for managers, human resources and occupational health teams, equality, diversity and inclusion leads, trade unions and employees.
Its four modules cover menopause awareness, symptom management, menopause champion training and cognitive behavioural strategies.
These are techniques that help people spot and change thought or behaviour patterns that can make symptoms harder to manage.
Amanda Griffiths, emeritus professor of occupational health psychology at the University of Nottingham, said: “I am so pleased the Wellbeing of Women’s toolkit is now available.
“It presents the culmination of many years’ research by Claire Hardy, Myra Hunter and myself at our three universities.
“It’s a clear story: women represent nearly half the working population and menopause is a normal event in their lives.
“Those who experience difficulties appreciate understanding and support. And it’s not difficult to provide.
“I really hope that the next generation of working women, their colleagues and their managers will find the toolkit helpful.”
MENO-Kit was informed by research studies conducted by Lancaster University, the University of Nottingham and King’s College London.
This included the UK’s first large-scale study of women’s experience of menopause at work, carried out at the University of Nottingham, which identified fatigue, poor concentration, low mood and hot flushes as symptoms affecting working life.
It also highlighted the kinds of employer support working women said they would find helpful.
The toolkit has been tested in eight UK organisations across the public, private and charity sectors.
A total of 2,162 people, mostly women in their mid-40s and 50s, contributed across the studies.
The launch comes at a time of growing urgency for employers.
Research from McKinsey & Company highlights the economic opportunity of closing the women’s health gap, estimating it could unlock up to £11bn a year for the UK economy.
The NHS Confederation has also highlighted the economic case for investing in women’s health, linking better support to improved workforce retention and reduced pressure on employers and public services.
Alongside this, research has continued to show the impact of menopause at work.
Separate polling by Benenden Health found that 28 per cent of women had considered leaving work because of symptoms, while 31 per cent reported reduced productivity.
MENO-Kit has been developed to help organisations respond with practical, evidence-based tools that build confidence, reduce stigma, improve wellbeing and help retain experienced staff.
The launch is being marked by an online event featuring a keynote from the Rt Hon Dame Diana Johnson, minister of state for employment, a live demonstration of MENO-Kit by Dr Abigail Morris, and a discussion on practical approaches to creating menopause-supportive workplaces.
Janet Lindsay, chief executive at Wellbeing of Women, said: “Too many women still struggle in silence during menopause at work, with many employers lacking the tools to respond effectively.
“MENO-Kit addresses this gap, offering practical, evidence-based ways for organisations to better support their people.
“It helps women stay working, thrive, and realise their potential at work.
“We’re hugely grateful to the research teams whose expertise has made this possible.”
Dr Abigail Morris, lecturer in workplace health and wellbeing at Lancaster University, said: “We’re delighted to launch Meno-Kit which represents an important step forward in supporting organisations across the UK to take a proactive and informed approach to menopause in the workplace.
“By providing practical tools, evidence-based guidance and a structured framework for action, the resource will help organisations develop effective menopause action plans, foster more inclusive and supportive workplace cultures, and better support women experiencing menopause to remain healthy, engaged and thriving at work.
“We believe Meno-Kit has the potential to drive meaningful organisational change while improving the everyday working lives and wellbeing of women across the UK workforce.”
Davina McCall, Wellbeing of Women ambassador, said: “Menopause shouldn’t be something women hide or struggle with alone at work or anywhere.
“It’s a normal life stage, not a personal failing.
“By talking about menopause openly and putting the right support in place, workplaces can make a huge difference to women’s wellbeing, confidence and careers enabling them to thrive at work.”

 Menopause2 weeks ago
Menopause2 weeks agoPerimenopause misinformation ‘putting women at risk’

 Hormonal health3 weeks ago
Hormonal health3 weeks agoNHS urged to update website following renaming of PCOS

 Insight4 weeks ago
Insight4 weeks agoPCOS renamed after decade-long campaign to end ‘cyst’ misconception

 Menopause2 weeks ago
Menopause2 weeks agoWomen still being failed when they reach menopause, experts say

 Entrepreneur2 weeks ago
Entrepreneur2 weeks agoWomen’s Health Innovation Summit opens submissions for 2026 Innovation Showcase

 Diagnosis4 weeks ago
Diagnosis4 weeks agoArtera receives FDA Clearance for breast cancer platform

 News2 weeks ago
News2 weeks agoThree menopause innovators shortlisted for Femtech World Award

 Fertility4 weeks ago
Fertility4 weeks agoAI could transform ovarian care through personalisation, study finds

















