News
Start-up raises US$2m to develop “novel” contraceptive device
The device consists of three magnetic units that are inserted individually into the uterus and self-assemble into a triangle

The US clinical development company 3Daughters has raised over US$2m in funding to develop a “novel” contraceptive device.
Massachusetts-based 3Daughters has developed a frameless delivery platform, based on physics and geometry, for targeted therapy directly into the uterus. Its copper IUD is the first product resulting from the technology.
The device consists of three magnetic units that are inserted individually into the uterus and self-assemble into a triangle which delivers copper to the uterus for contraception. It is removed by using a retriever with a magnetic tip that attracts the units, making the removal less painful.
The system, 3Daughters argues, could be capable of eliminating a number of “pain points” that keep women from getting an IUD.
Commenting on the funding, Mary Beth Cicero, co-founder and CEO of 3Daughters, said: “We are thrilled to have garnered such strong support from our existing and new investors, all of whom share our vision of solving health issues for women.
“This investment marks the next chapter for 3Daughters and allows us to execute our streamlined development plans to move into the clinic. With this funding, we are getting closer to our promise to address significant and neglected issues, like IUD insertion pain, that women have been putting up with for over 50 years.”
She added:”We aim to bring in an additional US$1m to close the planned US$3m seed financing round by the first quarter of 2024.”
According to Cicero, proceeds from the funding, led by Thairm Bio with participation from the Argosy Foundation and Wexford Science and Technology, will be used to advance the device through an Investigational New Drug Application (IND) filing and a phase 1 clinical trial.
Mark Bamforth, founder of Thairm Bio, said: “The lack of innovation in women’s health and the significant need 3Daughters is addressing drew me to this investment. It is clear that women’s health gets overlooked from an investment perspective and I want to be a part in changing that dynamic.
“Given the apparent urgency for new and effective contraceptive options and the pain women experience with IUDs, I recognised the need and formed an investment vehicle with my colleagues to help fund this important innovation for women.”
Emily Van Dunk, executive director of the Argosy Foundation, said: “Supporting 3Daughters aligns with our vision of empowering people and making a positive impact on communities globally.
“The recent changes in reproductive rights have strengthened our resolve to continue funding access to contraceptives, and we continue to support advancing women’s health with our investment in 3Daughters.
“We believe in the transformative potential of the 3Daughters novel IUD to make a significant impact for women.”
Thomas Osha, executive vice president at Wexford Science & Technology, added: “We were attracted by the innovative technology of a frameless uterine delivery platform and the significant market need of addressing IUD insertion pain.
“3Daughters has an outstanding scientific advisory board and through this partnership, we will provide access to Wexford’s substantial network of university talent, industry experts and strategic partners, as well as resources to help them scale and grow even faster.
“The 3Daughters team’s commitment to innovation for women, solid operational, scientific and executive leadership, and, most importantly, the potential for positive impact on women’s health make this investment an exciting venture for us.”
To receive the Femtech World newsletter, sign up here.
Insight
British women among angriest in Europe, health survey reveals

British women are among the angriest in Europe, a global health survey has revealed.
More than 20 per cent of women in Britain said they had experienced feelings of rage for much of the previous day.
British women were also 47 per cent more likely to say they felt angry than a year earlier.
The findings were published in the Hologic Global Women’s Health Index, a yearly league table based on polling of more than 76,000 women and girls aged over 15 worldwide.
Anna O’Sullivan, co-founder of women’s health awareness group CensHERship and founder of the FutureFemHealth news platform, told the Daily Mail: “These figures reflect years of long waiting lists, delayed diagnoses and women’s health being treated as an afterthought.
“We’ve seen a significant increase in awareness and discussion about women’s health over the last few years, but access to care has not kept up with that.
“These findings should be a wake-up call that it’s time for long-term, sustainable investment to ensure women can access timely healthcare, trusted information and earlier diagnosis before conditions become more complex and costly to treat.”
The data suggested anger levels among British women have risen sharply.
Rates across the rest of Europe, however, remained broadly the same.
The survey, which involved more than 140 countries, found three in 10 UK women said they felt sadness, compared with the EU average of 25 per cent.
The data, collected in February 2024 and released this week, also showed that around four in 10 women in both the UK and EU felt worry.
A third of women in the UK reported being in pain, up 10 per cent on the previous year.
Three in 10 women also said they lived with chronic health problems, up seven per cent on the year before.
Chronic health problems are long-term conditions that may need ongoing care or management.
Health experts said women in the UK were increasingly frustrated by the gap between the NHS care they expected and the care they received.
The report took a snapshot of the national mood, with participants asked about the emotions they had experienced “during a lot of the day yesterday”.
The UK placed sixth among 37 European countries for anger.
The highest levels were recorded in Malta, where 26 per cent of women reported feelings of rage, followed by Greece at 25 per cent, the Czech Republic and Albania at 23 per cent, and Spain at 22 per cent.
Ireland ranked at 18 per cent, while Germany, France and Switzerland each reported 17 per cent.
Britain has also slipped in Hologic’s overall global rankings for women’s health.
The UK is now 48th, close to dropping out of the top third of countries worldwide, after ranking 40th out of 142 countries last year.
Taiwan ranked first, followed by Latvia, Japan, Vietnam and Poland. Singapore, Germany and Austria were also among the leading countries.
Tim Simpson, a senior manager at Hologic, said: “Women are telling us they want earlier diagnosis and faster access to care.
“Improving women’s health will take continued commitment from policymakers, the NHS, clinicians and industry working together to deliver the changes women are asking for.”
A separate Hologic survey carried out last month found that almost 70 per cent of women had faced delays seeking NHS care in the past five years.
Two in five said difficulties accessing healthcare had left them feeling frustrated or anxious.
The survey’s findings reinforced official figures showing that Britain has become more anxious since before the pandemic.
The Office for National Statistics said 22.5 per cent of UK adults reported “high anxiety yesterday” between July and September 2024, up from 20.4 per cent in the same period in 2019.
Among women, the figure was 26.3 per cent, compared with 18.5 per cent among men.
A Department of Health and Social Care spokesperson said: “It is unacceptable that the UK continues to lag behind other countries when it comes to women’s health.”
Insight
Women’s mental health happens between appointments
By Ritika Sukhani – Psychologist, part of Véa’s Clinical Advisory Board
Women’s mental health often unfolds before, between and around appointments, long before it can be neatly explained in one conversation.
A woman arrives at a GP appointment with six months of feeling “off” behind her. Not acutely unwell. Not necessarily in crisis. Just not herself.
Poor sleep. Brain fog. Lower tolerance. Irritability before her period. Anxiety before meetings. A sense that her cycle, workload, relationships and energy are interacting – but not in a way she can neatly explain when the appointment begins.
She has tracked symptoms, Googled at midnight, screenshotted articles, made notes in her phone and tried to remember what changed, when it changed and what might have triggered it.
But when she finally gets the chance to talk, the task is not only to describe how she feels. It is to organise months of fluctuating experience into a story clear enough to communicate.
This is one of the most overlooked forms of labour in women’s mental health: not simply experiencing distress but having to make it coherent.
In the NHS, women’s mental health rarely arrives through one door. It may appear in primary care as exhaustion, poor sleep or “not feeling like myself”.
It may appear in NHS Talking Therapies as anxiety or low mood. It may sit beneath a menopause conversation, a fertility journey, chronic pain, trauma, caring responsibilities, burnout or the quiet work of holding everyone else together.
The NHS provides essential care.
NHS Talking Therapies received 1.81 million referrals in 2024/25, with 1.21 million referrals accessing services and 50.5 per cent of referrals moving to recovery after completing treatment.
But women’s lives do not unfold in pathway-shaped ways.
Their distress is often cumulative, contextual and relational. It is shaped not only by symptoms but by the conditions around those symptoms: work, debt, trauma, hormones, caregiving, discrimination, physical health, relationships, sleep and the pressure to keep functioning.
That is why women’s mental health needs more than access to services.
It needs continuity.
The work before the appointment
Women are often encouraged to advocate for themselves in healthcare. At its best, this is empowering. It supports agency, preparation and active participation in care.
But self-advocacy can also become another unpaid job.
It requires women to notice what is happening, remember when it started, connect it to context, decide what feels relevant, find the right words and communicate it clearly – often while already tired, anxious, in pain or emotionally overwhelmed.
In clinical care, distress is often the beginning of an assessment. Clinicians are trained to explore duration, severity, functioning, risk, context, history and meaning.
That process matters. It is how distress becomes understood with care.
The issue is broader than any single consultation.
Women’s mental health experiences often unfold over weeks, months and years, while healthcare conversations may happen in short, pressured windows.
Patterns form outside the appointment room: between cycle phases, work demands, caring responsibilities, relational stress, poor sleep and moments of emotional overload.
The UK Government’s Women’s Health Strategy survey found that 84 per cent of respondents said there had been times when they, or the woman they had in mind, were not listened to by healthcare professionals.
It points to something structural: the difficulty of making complex, fluctuating and context-dependent experiences legible inside systems that are often fragmented, time-limited and under pressure.
Endometriosis shows why continuity, language and recognition matter.
Research has found that delays in diagnosis can occur at both patient and medical levels, including when symptoms are normalised by women themselves and by doctors.
For many women, receiving a diagnosis did more than name pain; it provided language, reassurance and possible management strategies.
This is the point we should pay attention to.
Women do not always lack information. Often, they have fragments of it.
The challenge is how to preserve context, recognise recurrence and bring scattered experiences into a form that supports reflection, communication and earlier action.
Tracking captures moments. Continuity reveals patterns
We have made enormous progress in helping women track their bodies.
We can now log cycles, sleep, mood, fertility windows, temperature, symptoms, recovery and heart rate variability with increasing precision. Period-tracking apps, for example, have been described as tools that can support body awareness and menstrual health literacy, while also introducing new forms of work, distress and privacy concern for some users.
That tension matters.
Tracking can help women notice what is happening. But noticing is not the same as understanding. And understanding is not the same as being able to explain.
A cycle tracker can show when a period started. A wearable can show disrupted sleep. A symptom log can hold isolated data points. A notes app can capture fragments of a difficult week.
But unless those pieces are brought together, the interpretive work still falls to the woman.
She is left asking: Is this hormonal? Is this stress? Is this burnout? Is this anxiety? Is this normal for me? Is this pattern important? Should I mention it? How do I explain it?
This is where the first wave begins to meet its limits.
The first wave helped women capture signals.
The next wave could help women interpret context.
The promise of patient-generated health data has always been that it could bridge the gap between everyday life and formal care. But reviews continue to highlight challenges around integration with electronic health records, trust, provenance, data quality and contextual information.
That matters because women’s health data does not become useful simply because it is collected. It becomes useful when it is contextualised.
A poor night’s sleep means something different after one stressful day than after six weeks of overextension.
A low mood entry means something different when it appears in isolation than when it appears repeatedly around a cycle phase, a work pattern or a relationship dynamic.
A spike in anxiety means something different when it is viewed alongside workload, recovery, conflict, hormonal change or self-critical thinking.
The value is not in turning every experience into a metric.
The value is in seeing what repeats, how it repeats and what else is happening around it.
Women’s mental health is systemic
The latest Adult Psychiatric Morbidity Survey found that one in five adults in England had a common mental health condition, with prevalence higher in women at 24.2 per cent compared with 15.4 per cent in men.
It also found a clear socioeconomic gradient, with common mental health conditions more prevalent in the most deprived areas and among people with problem debt.
These figures matter because they remind us that women’s mental health cannot be understood only at the level of individual coping.
Of course, individual support matters. Therapy matters. Medication may matter. Assessment, formulation, risk management and diagnosis all have their place.
But a systemic lens asks what else is happening around the woman.
Who is she caring for? What is she carrying at work? What has she had to normalise? What physical symptoms have been separated from her emotional life? What inequalities shape how quickly she is heard, referred or supported? What happens while she is waiting?
Women’s mental health is often partly biological, partly psychological, partly relational, partly occupational and partly social.
It may not fit neatly into one symptom box at first. It may need time, pattern and context before it becomes clear what kind of support is needed.
This is why the missing layer is not more monitoring – it is supported sense-making.
The need for supported sense-making
Supported sense-making means moving beyond isolated logs and dashboards towards tools that help women understand how different parts of their lives interact over time: body, mood, cycle, stress, relationships, work, recovery and self-talk.
It also means being clear about what technology should and should not do.
Technology should not replace clinical assessment. It should not diagnose from a journal entry.
It should not turn ordinary emotion into pathology or place yet another responsibility on women to optimise themselves.
At its best, it can support the wider ecosystem around care.
It can help women hold onto context before an appointment.
It can help them notice patterns while they are waiting. It can support reflection between sessions. It can help them arrive at conversations with more continuity, while respecting the clinical judgement, formulation and relational care that services provide.
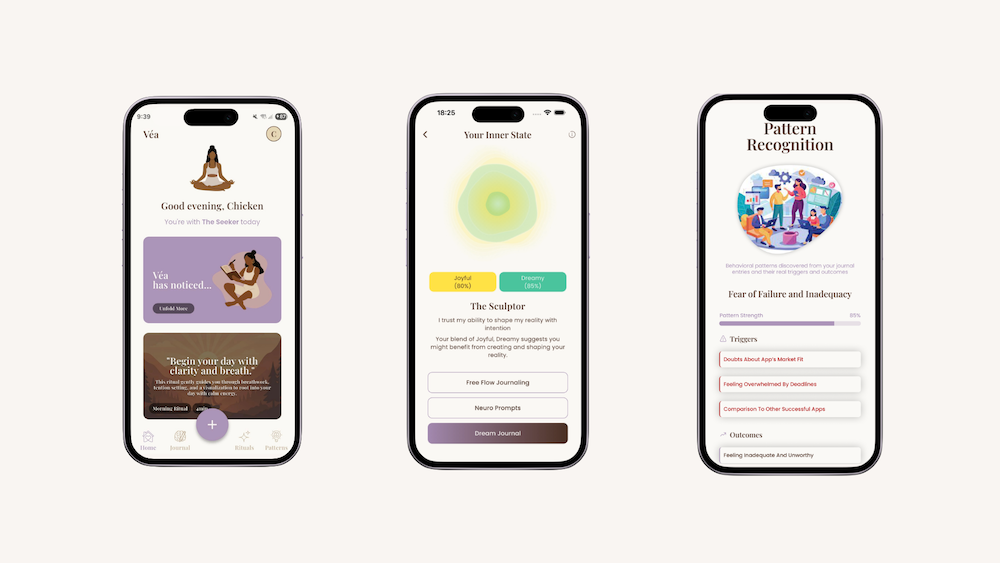
This is where platforms like Véa point toward a thoughtful role for women’s health technology.
By treating reflection, emotion and language as part of women’s longitudinal health context, Véa supports the kind of pattern recognition that often gets lost in daily life. Through micro-check-ins, journalling and reflective prompts, it helps women notice shifts in their internal state over time – not to self-diagnose but to build a clearer relationship with their own patterns.
For some women, that may support a better conversation with a clinician.
For others, it may help them recognise early signs of burnout, understand cyclical changes or notice when work, relationships and recovery are interacting in ways they had not previously named.
At its best, this kind of technology reduced the burden of self-interpretation. It helps women hold onto the thread.
From more data to better continuity
The women’s health gap is often discussed as a research gap, a funding gap and a diagnostic gap.
It is all of those things. McKinsey Health Institute and the World Economic Forum estimate that closing the women’s health gap could add at least $1 trillion annually to the global economy by 2040.6
But there is another gap sitting underneath the others.
A continuity gap.
Women’s mental health experiences are often long, fluctuating and context-dependent. Healthcare systems often encounter them in snapshots. Workplaces may only notice them when performance drops. Women themselves may only recognise the pattern once they are already exhausted.
That gap between lived experience and later explanation is where too much meaning gets lost.
The next generation of women’s health technology should help preserve the story behind the symptom.
It should make room for emotional, cognitive, hormonal and social context without collapsing everything into diagnosis or optimisation. It should support better conversations without pretending to replace clinical care.
Women should not have to rely on memory alone to make sense of months of fluctuating emotional, cognitive and physical experience.
The future of women’s mental health will not be built by asking women to monitor themselves more closely.
It will be built by helping them understand themselves sooner – and arrive at conversations about their health with more continuity, context and clarity.
Learn more about Véa at veajournal.app
Cancer
Life-prolonging ovarian cancer drug approved for use in England

 Menopause1 week ago
Menopause1 week agoPerimenopause misinformation ‘putting women at risk’

 News4 weeks ago
News4 weeks agoNIH Grant terminations disproportionately impact minority scientists, research finds

 Insight3 weeks ago
Insight3 weeks agoPCOS renamed after decade-long campaign to end ‘cyst’ misconception

 Adolescent health4 weeks ago
Adolescent health4 weeks agoWUKA brings Period-Positive Pool Party to London Aquatics Centre to keep girls swimming through puberty

 Hormonal health2 weeks ago
Hormonal health2 weeks agoNHS urged to update website following renaming of PCOS

 Menopause4 weeks ago
Menopause4 weeks agoCBT shows promise for menopause insomnia and hot flashes

 Entrepreneur1 week ago
Entrepreneur1 week agoWomen’s Health Innovation Summit opens submissions for 2026 Innovation Showcase

 News1 week ago
News1 week agoThree menopause innovators shortlisted for Femtech World Award




























2 Comments